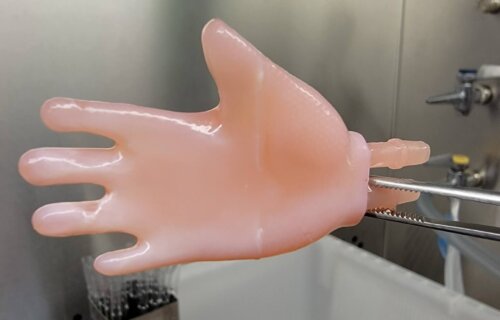
NEW YORK — Bioengineers in New York are working on a game-changing method to grow skin in the shape of complex body parts — like a hand. The innovation would help surgeons grafting artificial skin onto injured patients. Researchers at Columbia University say engineered skin typically comes in flat pieces. Like wrapping paper, this makes it difficult and time-consuming to stitch it together around an irregularly-shaped limbs. The new method solves this problem by growing engineered skin in complex, three-dimensional shapes ahead of time.
It would make it possible to construct, for example, a seamless “glove” of skin cells that doctors can be easily slip onto a severely burned hand.
“Three-dimensional skin constructs that can be transplanted as ‘biological clothing’ would have many advantages,” says lead developer Hasan Erbil Abaci, PhD, an assistant professor of dermatology at Columbia’s Vagelos College of Physicians and Surgeons, in a university release. “They would dramatically minimize the need for suturing, reduce the length of surgeries, and improve aesthetic outcomes.”
The current study also demonstrates that continuous 3D grafts have better mechanical and functional properties than conventional grafts which surgeons have to piece together.
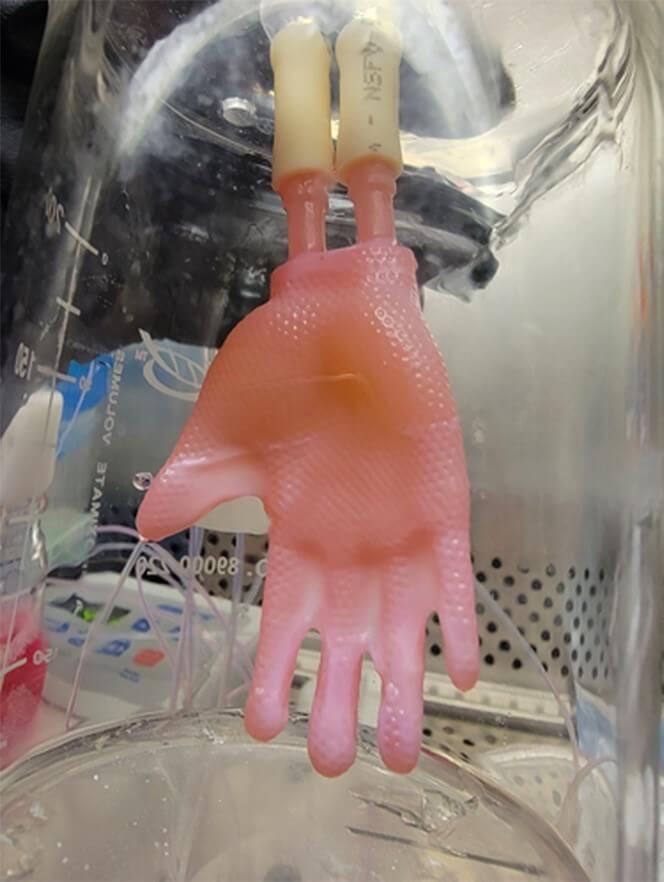
How did scientists build the hand?
The process of creating the new skin grafts begins with a 3D laser scan of the target structure, such as a human hand.
Next, researchers craft a hollow and permeable model of the hand using computer-aided designs and 3D printing. Scientists then seed the exterior with skin fibroblasts, which generate the skin’s connective tissue and collagen — a structural protein. Finally, the team coats the outside of the mold with a mixture of keratinocytes, which are cells that make up most of the outer skin layer, or epidermis. The inside is perfused with growth media, which supports and nourishes the developing graft.
Except for the 3D scaffold, the researchers employed the same procedures which make flat engineered skin. The entire process takes roughly the same amount of time, about three weeks. In a first test of the 3D engineered skin, the team grafted these human skin cell constructs onto the hind limbs of mice.
“It was like putting a pair of shorts on the mice,” Abaci says, “The entire surgery took about 10 minutes.”
One month later, the grafts completely integrated with the surrounding mouse skin, and the mice displayed full function of their limbs. Researchers note that mouse skin heals differently than human skin. Next, they plan to test the grafts on larger animals with skin biology that more closely resembles humans. However, clinical trials on humans are likely years away.
A tiny amount of skin is all scientists need
The 3D grafts are the first major re-design of engineered skin grafts since scientists first created them in the early 1980s.
“Engineered skin started with only two cell types, but human skin has around 50 types of cells. Most research had focused on mimicking the cellular components of human skin,” Abaci continues. “As a bioengineer, it’s always bothered me that the skin’s geometry was overlooked and grafts have been made with open boundaries, or edges. We know from bioengineering other organs that geometry is an important factor that affects function.”
Abaci and his team realized they could make more lifelike grafts after the invention of 3D printers.
“We hypothesized that a 3D fully enclosed shape would more closely mimic our natural skin and be stronger mechanically, and that’s what we found,” Abaci says. “Simply remaining faithful to the continuous geometry of human skin significantly improves the composition, structure, and strength of the graft.”
In the future, Abaci envisions grafts that are custom-made from a patient’s own cells. Using a 4X4-millimeter skin sample, researchers say would have enough cells to create a culture and multiply it to create enough skin to cover a full-size human hand model.
“Another compelling use would be face transplants, where our wearable skin would be integrated with underlying tissues like cartilage, muscle, and bone, offering patients a personalized alternative to cadaver transplants,” the researcher concludes.
The researchers published their findings in Science Advances.
South West News Service writer Dean Murray contributed to this report.