CHICAGO — An implant has been developed that relieves pain without the need for prescription drugs. The dissolvable device works by softly wrapping itself around nerves and delivers precise cooling by numbing them, blocking signals to the brain. An external pump enables remote activation by the patient, rising or reducing intensity. Once no longer needed the bioelectronic tool is absorbed naturally by the body.
Researchers hope the first-of-its-kind device will put a dent in the opioid crisis that continues to grip America. Giving patients an effective option that doesn’t run the risk of addiction to prescription drugs — nor any side effects from such meds — would certainly be a welcome alternative for individuals prone to dependence.
“Although opioids are extremely effective, they also are extremely addictive,” says project leader John Rogers, a professor of materials science and engineering, biomedical engineering and neurological surgery at Northwestern University, in a statement. “As engineers, we are motivated by the idea of treating pain without drugs – in ways that can be turned on and off instantly, with user control over the intensity of relief.
“The technology reported here exploits the mechanism that causes your fingers to feel number when cold,” he continues. “Our implant demonstrates in animal model studies that this effect can be produced in a programmable way, directly and locally to targeted nerves – even those deep within surrounding soft tissues.”
Pain-relief implant could especially useful after surgery
The novel instrument is small and flexible. It could potentially replace post-operative medications. Researchers believe it will be most valuable after routine surgeries — or even amputations. Surgeons would insert it during the procedure. The patient would then be able to manage their pain afterwards.
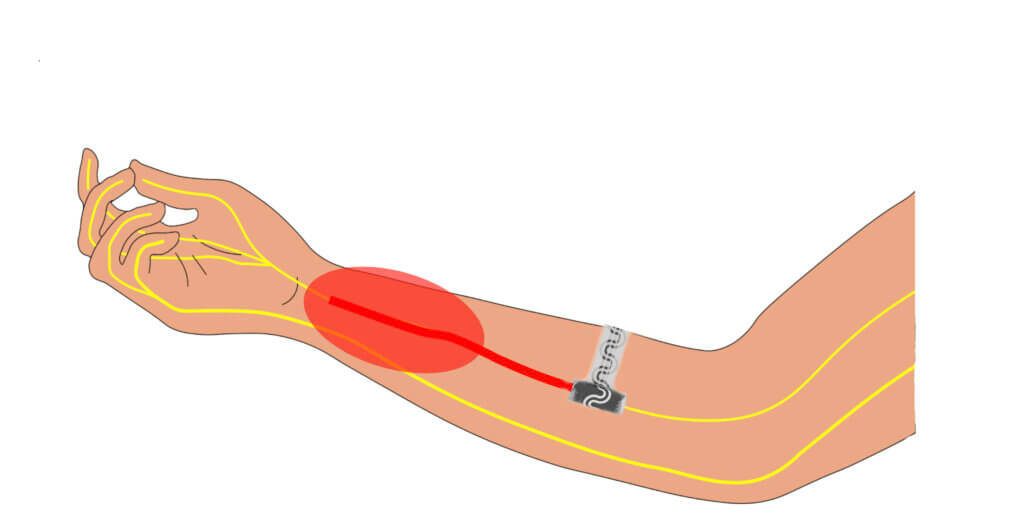
A liquid called perfluoropentane is induced in a tiny channel to evaporate at the specific location of a sensory nerve, similar to how sweat cools the body. It is already clinically approved as an ultrasound contrast agent and for pressurized inhalers.
“As you cool down a nerve, the signals that travel through the nerve become slower and slower – eventually stopping completely,” explains co-author Dr. Matthew MacEwan, of Washington University in St Louis. “We are specifically targeting peripheral nerves, which connect your brain and your spinal cord to the rest of your body. These are the nerves that communicate sensory stimuli – including pain. By delivering a cooling effect to just one or two targeted nerves, we can effectively modulate pain signals in one specific region of the body.”
A second channel contains dry nitrogen. When the liquid and inert gas flow into a shared chamber, the desired reaction occurs. A tiny integrated sensor monitors the temperature of the nerve to ensure it is not getting too cold.
“Excessive cooling can damage the nerve and the fragile tissues around it. The duration and temperature of the cooling must therefore be controlled precisely,” says Rogers. “By monitoring the temperature at the nerve, the flow rates can be adjusted automatically to set a point that blocks pain in a reversible, safe manner. Ongoing work seeks to define the full set of time and temperature thresholds below which the process remains fully reversible.”
Device simply ‘melts into bodily fluids’ eventually
Previous attempts to block nerves with cryotherapies injected with a needle have been less successful. Imprecise approaches cool large areas – increasing the risk of inflammation and other side effects.
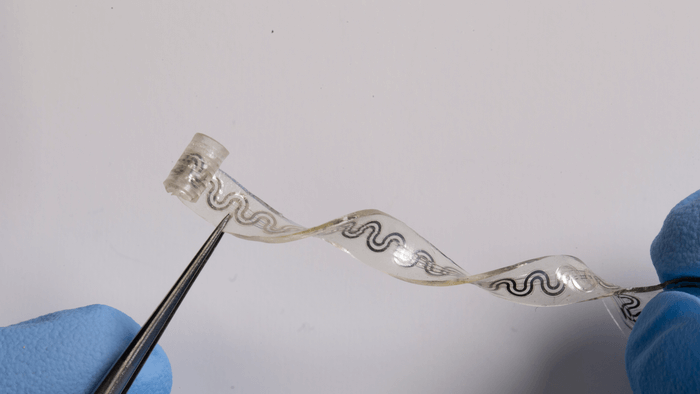
The device is as thin as a piece of paper, at just 5 millimeters in diameter at its widest point. It is similar to absorbable stitches, melting into bodily fluids in days or weeks. The elasticity makes it ideal for treating highly sensitive nerves.
“If you think about soft tissues, fragile nerves and a body that’s in constant motion, any interfacing device must have the ability to flex, bend, twist and stretch easily and naturally,” says Rogers. “Furthermore, you would like the device to simply disappear after it is no longer needed, to avoid delicate and risky procedures for surgical removal.”
One end is curled into a cuff that softly wraps around a single nerve, bypassing the need for sutures. By targeting only the affected nerve, it spares surrounding regions from unnecessary cooling.
“You don’t want to inadvertently cool other nerves or tissues unrelated to the nerve transmitting the painful stimuli,” explains MacEwan. “We want to block the pain signals, not the nerves that control motor function and enables you to use your hand, for example.”
Zapping painful stimuli with electrodes have also been found to have several limitations.
“You can’t shut down a nerve with electrical stimulation without activating it first,” adds MacEwan. “That can cause additional pain or muscle contractions and is not ideal, from a patient’s perspective.”
South West News Service writer Mark Waghorn contributed to this report.
I’ve had hundreds of “absorbable stitches”. They don’t all melt into the body… having had to remove about 40 or so myself. Up to two years later you can find part of a stitch sticking up out of the skin, which is painful because it catches on clothing.