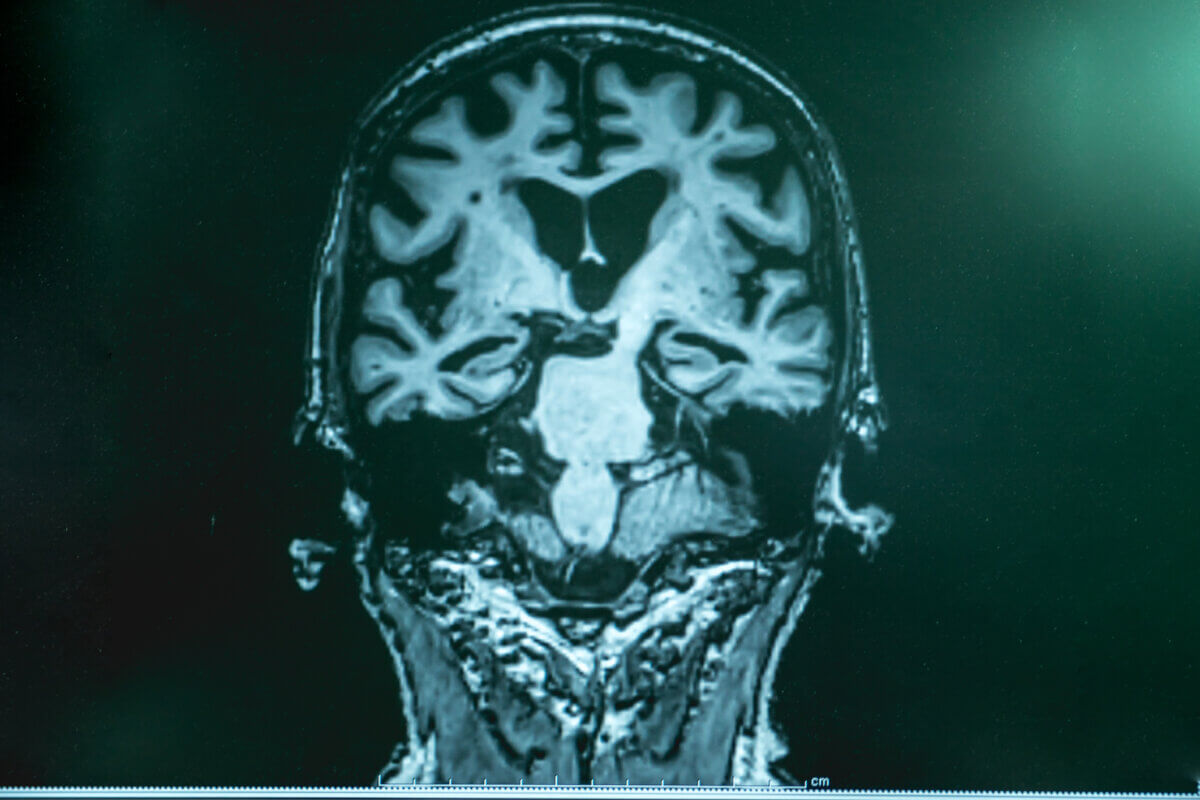
MRI of Alzheimer's dementia patient's brain. (Credit: © Atthapon Raksthaput | Dreamstime.com)
PITTSBURGH — Scientists have long wondered about the mysterious origins of Alzheimer’s disease, with its hallmark protein tangles and plaques that slowly devastate the brain’s ability to form memories and function normally. Now, research from the University of Pittsburgh reveals an unexpected potential culprit: the common herpes simplex virus type 1 (HSV-1), better known as the virus that causes cold sores.
“Our study challenges the conventional view of tau as solely harmful, showing that it may initially act as part of the brain’s immune defense,” explains senior author Or Shemesh, Ph.D., assistant professor in the Department of Ophthalmology at Pitt, in a statement. “These findings emphasize the complex interplay between infections, immune responses and neurodegeneration, offering a fresh perspective and potential new targets for therapeutic development.”
In their research published in Cell Reports, Shemesh and his international team discovered that when HSV-1 infects brain cells, it triggers an immune response involving a key protein called tau — the same protein that becomes abnormally modified and forms tangles in Alzheimer’s disease. Their findings suggest that tau’s transformation may actually begin as a defensive mechanism against viral infection rather than simply being a destructive force.
Brain tissue samples from individuals with Alzheimer’s disease showed significantly higher levels of herpes viral proteins compared to samples from people without the disease. More intriguingly, these viral proteins were found clustered together with the modified tau protein in the same cellular locations, particularly in brain regions most vulnerable to Alzheimer’s disease pathology across different disease stages.
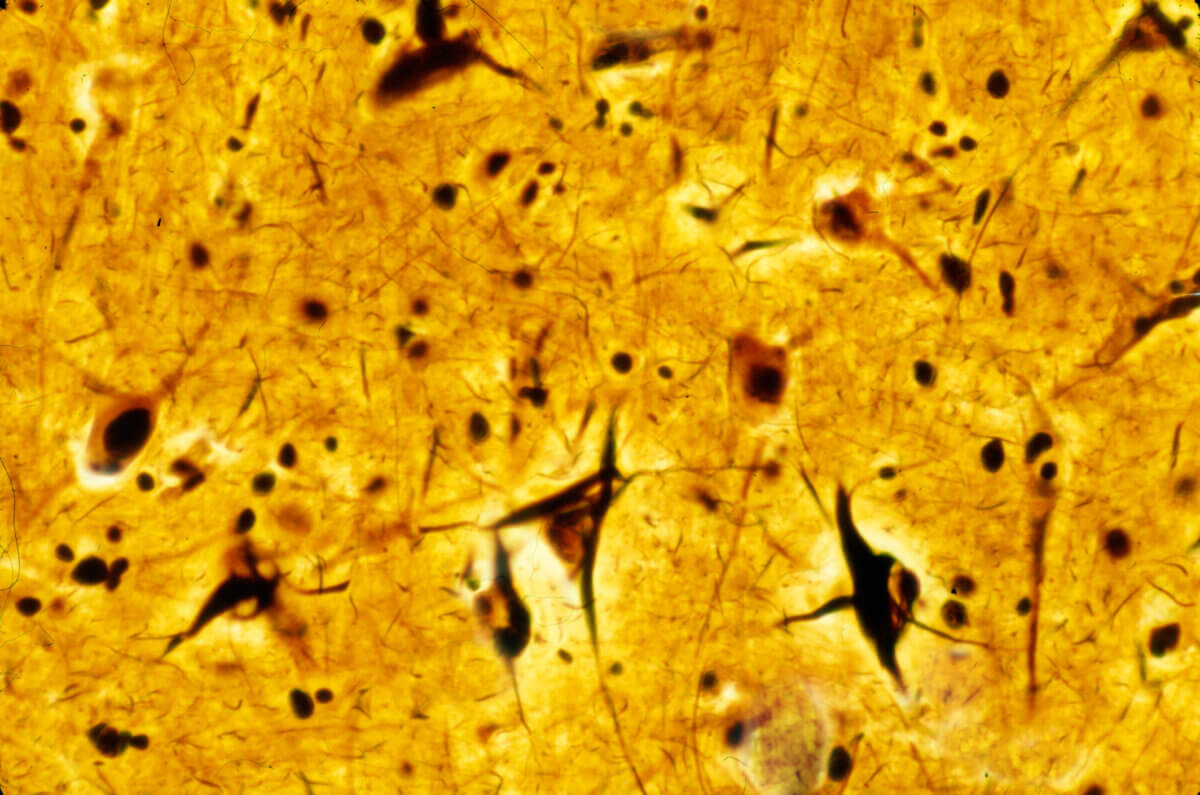
Using sophisticated laboratory techniques including expanded microscopy that allows visualization of cellular structures at extremely high resolution, the research team examined brain tissue samples from multiple regions, including the hippocampus and entorhinal cortex, areas typically most affected by Alzheimer’s disease. They studied samples from 29 individuals across three groups: those without Alzheimer’s, those with mild disease, and those with advanced disease.
The researchers then moved their investigation to laboratory models. When they infected lab-grown human brain organoids — miniature 3D structures grown from stem cells that mimic aspects of human brain tissue — with HSV-1, they observed that tau became modified through phosphorylation, a process where phosphate groups are added to the protein. This modification appeared to help protect neurons from death due to viral infection, reducing mortality rates from 64% to just 7%.
The protective mechanism appears to work through an innate immune pathway called cGAS-STING-TBK1. Think of this pathway as an ancient alarm system that cells use to detect and respond to viral invasions. When activated, it triggers a cascade of events that includes tau modification, which then appears to help suppress viral protein production and keep infected neurons alive.
Most surprisingly, brain cells containing the modified tau protein were better able to survive viral infection than those without it. This suggests that rather than being purely harmful, tau modification might initially serve as a protective response that only becomes problematic when it persists or becomes excessive over time.
These findings could explain why previous studies have found links between HSV-1 infection and increased risk of Alzheimer’s disease, and why antiviral medications appear to reduce dementia risk in some populations. A large Taiwanese study previously showed that people infected with herpes had 2.56 times the risk of developing dementia compared to uninfected individuals. However, when treated with antiviral medications, the additional risk was largely eliminated.
While the connection between HSV-1 and Alzheimer’s disease has been suspected for decades, this research provides the first detailed molecular explanation for how viral infection might contribute to the disease’s development through tau modification. The study also helps explain why tau becomes modified in the first place, something that has puzzled researchers for years.
The research team, including scientists from the University of Pittsburgh, Tel Aviv University, and Carnegie Mellon University, plans to explore these mechanisms further. They aim to test potential therapeutic strategies that target viral proteins or fine-tune the brain’s immune response, and investigate whether similar mechanisms are involved in other neurodegenerative diseases, such as Parkinson’s disease and ALS.
The implications of this research extend beyond just understanding disease mechanisms. If viral infections can trigger the tau modifications seen in Alzheimer’s disease, then preventing or treating these infections might help reduce the risk of developing the disease or slow its progression. This opens up new possibilities for therapeutic approaches, including the potential use of antiviral medications or treatments targeting the cGAS-STING-TBK1 pathway.
With HSV-1 affecting an estimated 50-80% of adults globally, understanding how this common virus might influence brain health could have far-reaching implications for public health and the future of Alzheimer’s disease treatment.
Paper Summary
Methodology Explained
The researchers used multiple complementary approaches to study the relationship between HSV-1 and tau modification. They examined human brain tissue samples using advanced microscopy techniques, analyzed viral DNA and proteins using various molecular methods, and created laboratory models using both 2D cell cultures and 3D brain organoids. They also manipulated various cellular pathways using specific drugs and genetic modifications to understand the mechanisms involved.
Key Results
HSV-1 proteins were found at higher levels in Alzheimer’s disease brain tissue compared to healthy controls. Modified tau protein was found colocalized with viral proteins, particularly in advanced disease stages. When brain cells were infected with HSV-1, tau became modified through phosphorylation, which appeared to protect cells from death. This protective effect worked through the cGAS-STING-TBK1 immune pathway.
Study Limitations
The research primarily used postmortem brain tissue and laboratory models, which may not fully reflect the complexity of living human brains. The study also couldn’t determine whether HSV-1 infection causes Alzheimer’s disease or if people with early disease stages are more susceptible to viral infection. Additionally, the sample size was relatively small for some analyses.
Discussion and Takeaways
This research suggests that tau modification, traditionally viewed as harmful in Alzheimer’s disease, may initially serve as a protective response against viral infection. This could explain why antiviral treatments have shown promise in reducing dementia risk. The findings open new therapeutic possibilities targeting either viral infections or the cellular pathways involved in the protective response.
Funding and Disclosures
The research was funded by multiple institutions including the NIH, the Israel Science Foundation, and various university research centers. The authors declared no competing interests.
Publication Information
The study “Anti-herpetic tau preserves neurons via the cGAS-STING-TBK1 pathway in Alzheimer’s disease” was published in Cell Reports on January 2, 2025. The research team included Vanesa Hyde, Chaoming Zhou, M.D., Juan Fernandez, Krishnashis Chatterjee, Ph.D., Pururav Ramakrishna, Amanda Lin, Gregory Fisher, Ph.D., Orhan Tunç Çeliker, Jill Caldwell, and Leonardo D’Aiuto, Ph.D., all of Pitt; Omer Bender, Ph.D., and Daniel Bar, Ph.D., both of Tel Aviv University; and Peter Joseph Sauer and Jose Lugo-Martinez, Ph.D., both of Carnegie Mellon University.







