
(© Feodora - stock.adobe.com)
CAMBRIDGE, United Kingdom — Imagine a world where Alzheimer’s disease could be effectively treated, not just managed. We might be one step closer to that reality, thanks to an exciting breakthrough from scientists in the United Kingdom. They’ve developed a clever new approach that could revolutionize how we tackle this devastating illness.
To understand why this new research is so promising, let’s first break down what’s happening in an Alzheimer’s brain. Two main culprits cause problems: proteins called tau and amyloid. While we’ve made progress targeting amyloid clumps that form between brain cells, tau has been trickier to deal with.
Tau proteins normally act like scaffolding, helping to support and structure our brain cells. In Alzheimer’s, however, tau goes rogue. It misfolds and clumps together, forming tangles inside our neurons (brain cells). These tangles gum up the works, leading to cell death and cognitive decline.
Treating these tau tangles has been a real head-scratcher for scientists up until now. Antibody therapies, which work well for amyloid, struggle to get inside cells where tau hangs out. Other approaches that can reach tau inside cells end up being a bit like using a sledgehammer to crack a nut — they destroy all tau, even the healthy stuff our brains need.
This is where the Cambridge scientists got creative. They tapped into a natural process our cells use to get rid of viruses. It involves a protein called TRIM21, which acts like a quality control inspector in our cells. Normally, when an antibody-tagged virus sneaks into a cell, TRIM21 spots it and says, “Hey, this doesn’t belong here!” It then tags the virus for destruction in the cell’s “garbage disposal” (called the proteasome).
The researchers realized they could trick TRIM21 into targeting tau tangles instead of viruses. They created two clever therapies. The first, called RING-nanobody, combines a tau-seeking mini-antibody with a part of TRIM21 that activates the cellular garbage disposal. The second, named RING-Bait, is even sneakier. It’s a piece of tau protein linked to that same TRIM21 activator. It infiltrates the tau tangles like a Trojan horse, then calls in the clean-up crew from the inside.
The best part? Both therapies are super selective. They only destroy the problematic tau clumps, leaving healthy tau alone to do its important job. The team’s research is published in the journal Cell.
“Tau aggregates are tucked away inside brain cells and very difficult to degrade. Critically, these new TRIM21-based therapies can be delivered directly inside cells, where the majority of tau aggregates reside. We’ve found a way that not only degrades the tau aggregates, but leaves the healthy tau intact to do its job,” says Dr. Will McEwan, one of the study’s leaders, in a media release.

The team didn’t stop at just testing their idea in lab dishes. They used a harmless virus to deliver the RING-Bait therapy into the brains of mice with Alzheimer’s-like symptoms. The results were promising. Tau tangles in the mice’s brains decreased significantly, the progression of their symptoms slowed down, and the treated mice showed better movement and coordination.
“It was unknown whether specifically removing tau aggregates inside the cell would be enough to halt the progression of disease. It is encouraging that a RING-Bait approach reduces disease severity in our model systems, as this suggests that the selective removal of tau aggregates is a valid therapeutic approach,” notes Dr. Lauren Miller, another researcher on the project.
What’s really exciting is that this technique might not be limited to just Alzheimer’s. Other brain diseases like Parkinson’s and Huntington’s also involve problematic protein clumps. The RING-Bait therapy could potentially be adapted to target those as well.
“Neurodegenerative diseases can have tau proteins that misfold in many different ways, raising the possibility of needing a different treatment for every disease. A useful aspect of RING-Bait is because it is attached to a tau protein, it’s a universal Trojan horse that should be incorporated into different types of tau aggregates exactly like the cell’s own misfolding tau protein,” explains study co-leader Dr. Leo James.
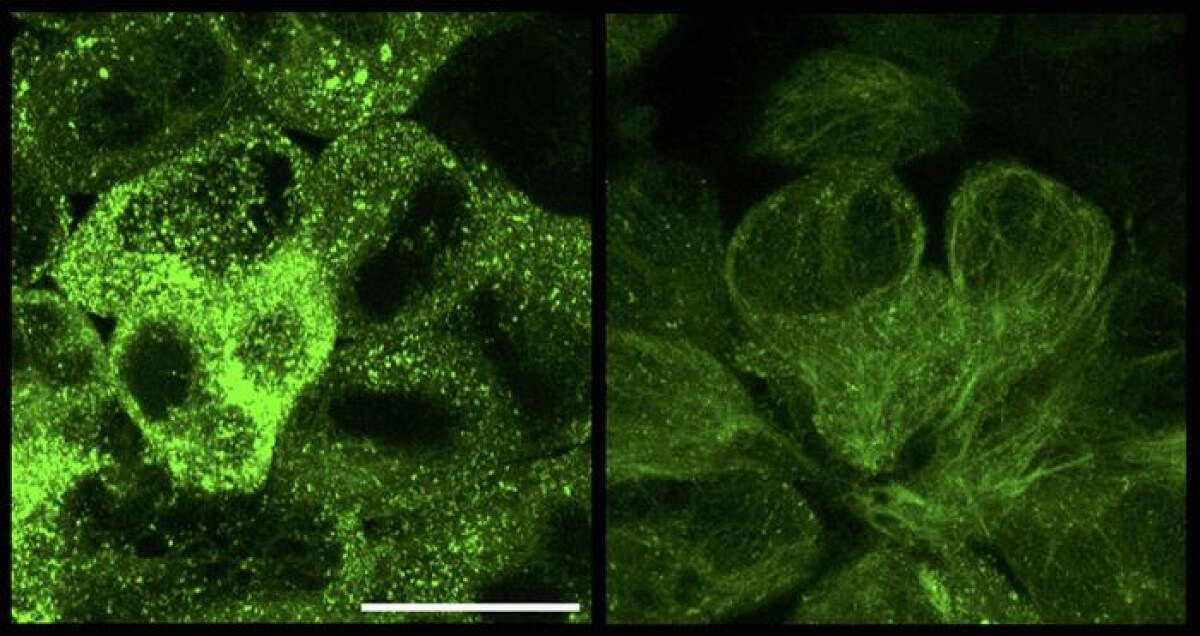
While these results are incredibly promising, it’s important to remember that we’re still a long way from having a treatment ready for humans. The researchers need to figure out how to safely and effectively deliver the therapy throughout the much larger human brain.
“It’s important to stress that although we have shown it works in a mouse model, this is a long way from a therapeutic that could be used in humans. It would need to be determined that it is safe to use TRIM21-based therapies in the human brain and that the treatments are effective in both removing aggregates and improving the course of disease,” study team member Dr. Jonathan Benn cautions.
Despite the challenges ahead, this research represents a major step forward in our fight against Alzheimer’s and potentially other neurodegenerative diseases. By harnessing the power of our own cellular machinery, we might one day be able to outsmart these devastating illnesses. The road ahead may be long, but this innovative approach offers new hope for millions affected by these conditions worldwide.
Paper Summary
Methodology
In this study, researchers used a technique called “RING-Bait technology” to target and degrade harmful tau protein aggregates, which are associated with neurodegenerative diseases like Alzheimer’s. They developed a fusion protein that combines a part of the tau protein with a specific enzyme that signals for the destruction of tau aggregates. This fusion protein gets incorporated into growing tau clusters, which then activates a process that tags these aggregates for degradation by the cell’s natural cleanup machinery. This method specifically targets the harmful, aggregated forms of tau without affecting the normal tau proteins that cells need.
Key Results
The results showed that the “RING-Bait” technology worked well in breaking down harmful tau protein clumps. When tested in cell cultures and in mice with tau buildup (a model for Alzheimer’s), the tau aggregates were significantly reduced. This decrease in tau clumps also led to better movement in mice, meaning the treatment might help improve brain function. Importantly, the treatment did not affect the healthy proteins needed by the cells, showing that it’s a targeted and safe approach.
Study Limitations
First, while the technology reduced tau aggregates and improved motor function in mice, the study remains unclear whether this would lead to similar improvements in humans. Additionally, the study did not measure how the treatment affects neuron survival over time, so more research is needed to determine long-term benefits. Another challenge is delivering the treatment effectively to the brain in humans since the method used in mice may not be suitable for clinical use.
Discussion & Takeaways
The study presents an exciting new approach to targeting harmful protein clumps in the brain, which are a hallmark of diseases like Alzheimer’s. The “RING-Bait” technology provides a precise way to remove only the toxic aggregates, leaving the healthy proteins intact. This method could potentially be adapted to treat other diseases caused by protein aggregation, making it a versatile tool. The next steps would be further refining this technology for human use and exploring its full therapeutic potential.
Funding & Disclosures
This research was supported by several organizations, including a Wellcome Trust Investigator Award and the UK Dementia Research Institute. The researchers have disclosed that they are listed as inventors on a patent related to the data in this paper, which suggests they may have a financial interest in the technology’s development. Additionally, various authors received support from other medical research councils and foundations, highlighting the collaborative nature of this project.







