
(© Creative Cat Studio - stock.adobe.com)
In A Nutshell
- Atorvastatin directly binds to muscle proteins — Scientists captured the first atomic-level images showing how the cholesterol drug latches onto calcium channels in muscle cells, forcing them open and causing calcium to leak out.
- Genetic mutations amplify the risk — People with certain RyR1 gene variants linked to malignant hyperthermia are significantly more vulnerable to statin-induced muscle damage because their calcium channels are already primed to malfunction.
- Your heart appears naturally protected — The heart’s version of the calcium channel has key structural differences that prevent atorvastatin from binding, which may explain why cardiac side effects are rare despite muscle complaints being common.
- Better drugs may be on the horizon — The research reveals exactly where the drug grabs onto muscle tissue versus where it blocks cholesterol production, providing a roadmap for designing statins that work without causing muscle pain.
For years, doctors have heard the same complaint from patients on statins: their muscles hurt. Some describe general achiness, others struggle with weakness, and in rare cases, people end up in the hospital with severe muscle breakdown. Now scientists have finally caught the culprit in action.
Atorvastatin, sold under the brand name Lipitor and taken by millions of Americans daily, is prescribed for lowering cholesterol. But it also latches onto proteins in muscle cells and forces open tiny doors that aren’t supposed to be open, causing calcium to leak out like water from a faulty dam. That leak may be one reason muscles hurt and, in some people, get seriously damaged.
The discovery, described in a pre-print paper ahead of publication in Nature Communications, provides direct evidence that these widely prescribed drugs can act on muscle tissue itself, settling uncertainty about whether the effects were direct or indirect.
Between 10 and 25 percent of people taking statins report muscle problems. Most push through mild discomfort, but some have to stop taking the medication entirely. A small number develop a life-threatening condition where muscle tissue breaks down so severely it can damage the kidneys.
A Molecular Crime Scene
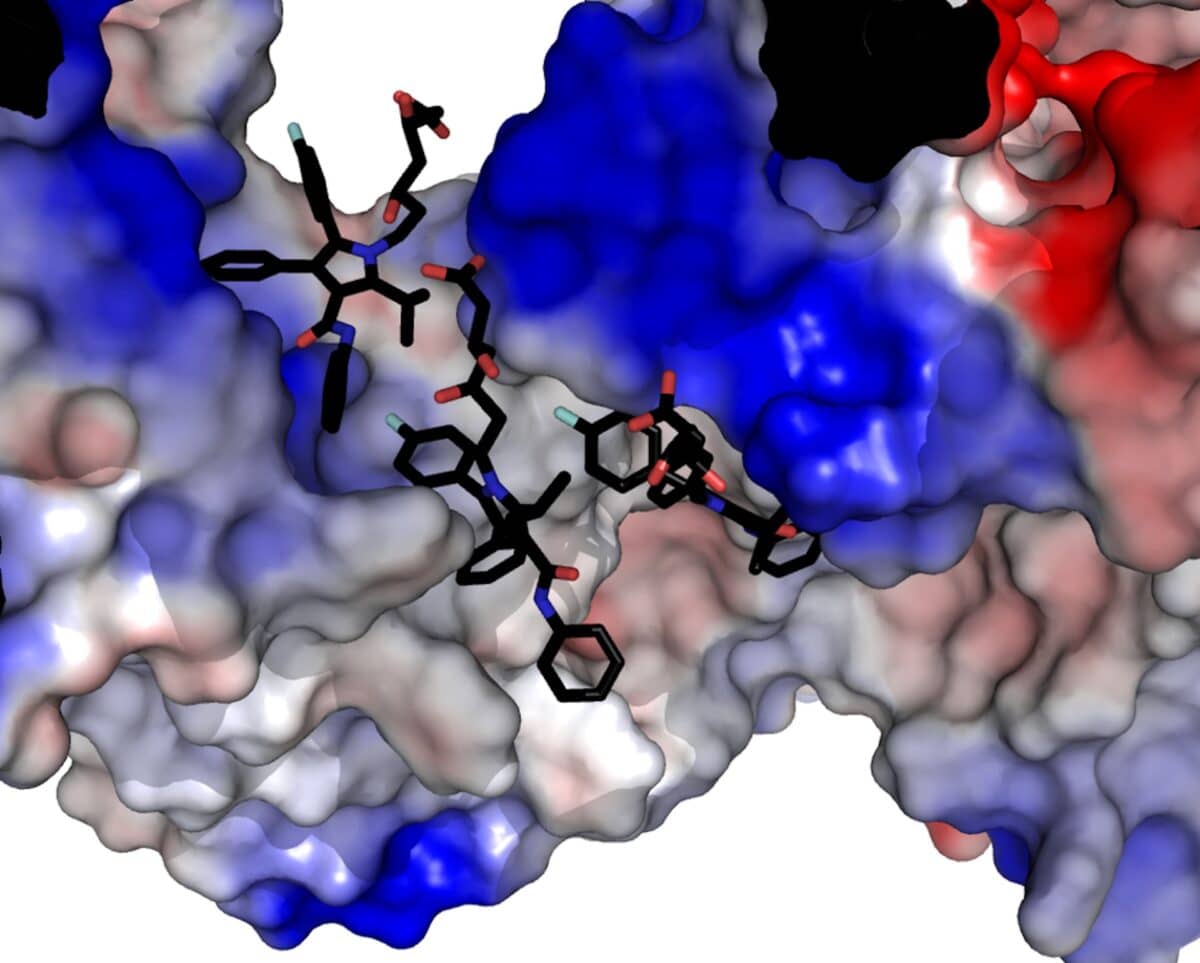
The research team used powerful microscopes to zoom in on the scene of the crime, capturing images detailed enough to see individual drug molecules grabbing onto a protein called RyR1. This protein normally acts as a carefully controlled gate, releasing bursts of calcium inside muscle cells to make them contract when you move.
But when atorvastatin shows up, everything changes. One drug molecule slips into a crack in the protein’s structure, nudging it slightly open. Then two more molecules pile on, clustering together and wedging the gate even wider. With the door stuck open, calcium leaks out continuously instead of coming out in controlled bursts.
The team from the University of British Columbia and University of Wisconsin-Madison tested the protein under normal conditions and found all the gates stayed properly closed. Add atorvastatin, and nearly two-thirds of them got jammed open.
The drug wasn’t designed to do this at all. Its job is to block an enzyme in the liver that makes cholesterol. Forcing open calcium gates in muscles is purely accidental collateral damage.
Some People Are More Vulnerable
Genetics plays a role in who gets hurt. Some people carry mutations in the gene that makes RyR1, and for them, the protein is already a bit unstable, easier to push open. When researchers tested one of these mutant versions with atorvastatin, more than 80 percent of the gates got stuck open, compared to about 64 percent for the normal version.
These mutations cause a rare but dangerous condition called malignant hyperthermia, where certain anesthesia drugs used during surgery trigger a catastrophic reaction. Several case reports have described people with these mutations who also had severe reactions to statins, and this study explains why: their calcium gates are sitting ducks, primed to malfunction.
The mutation sits far from where the drug binds, but it weakens the protein’s structure in a way that makes the whole system more fragile. It’s like having a door with loose hinges. Even a gentle push can force it open.
Why Your Heart Gets a Pass
Interestingly, hearts have their own version of this protein, called RyR2, and it’s very similar to the muscle version. Yet heart problems from statins are incredibly rare. Why do leg muscles cramp up while the heart keeps ticking along just fine?
The answer comes down to a few key differences in the protein’s structure. The spots where atorvastatin grabs hold of the muscle version don’t quite line up in the heart version. Some amino acids that help grip the drug in muscle are swapped out for different ones in the heart. One particular substitution would actually create a collision, physically blocking the drug from fitting in properly.
So the heart’s calcium gates appear naturally resistant to atorvastatin, which probably explains why people don’t develop dangerous heart rhythms as a side effect.
A Blueprint for Better Statins
By comparing how atorvastatin fits into its intended target (a liver enzyme) versus this accidental muscle target, researchers spotted an opportunity. The part of the drug that blocks cholesterol production sits deep inside the liver enzyme but barely touches the muscle protein.
What really grabs onto the muscle protein are the drug’s bulky ring-shaped structures, which stick out like handles when the drug is doing its proper job in the liver. Adding some extra chemical bulk to those rings could block them from fitting into the muscle protein while leaving the cholesterol-blocking function completely intact.
In other words, it might be possible to design statins that still lower cholesterol beautifully but don’t cause muscle pain.
Six different statins have all been shown to trigger this same problem with muscle proteins, though some are worse offenders than others. Four of them (cerivastatin, atorvastatin, fluvastatin, and rosuvastatin) share a particular chemical feature that makes strong contact with the muscle protein’s vulnerable spot.
The concentrations needed to cause trouble fall in the same range as levels seen in people on high doses, which suggests this effect could matter in real patients.
Interestingly, the same general region of the muscle protein where atorvastatin binds is also targeted by certain insecticides. These chemicals kill bugs by forcing open their version of these calcium gates. The insecticides are considered reasonably safe for people at typical exposure levels, but previous research found that people with those RyR1 mutations are more sensitive to them too.
It highlights that this particular region of the protein is a weak point, susceptible to being hijacked by various chemicals that were never meant to interact with it.
Takeaways For Patients Who Take Statins
This study provides the first rock-solid proof that statins physically bind to the muscle protein rather than causing problems indirectly through metabolism or other pathways. When doctors see elevated levels of certain muscle enzymes in the blood, it’s a sign of damage, and symptoms usually go away after stopping the medication.
The findings may help inform future work on whether genetic testing could identify people at higher risk before prescribing these drugs.
“Statins have been a cornerstone of cardiovascular care for decades,” said senior author Dr. Filip Van Petegem, a professor at the University of British Columbia’s Life Sciences Institute, in a statement. “Our goal is to make them even safer, so patients can benefit without fear of serious side effects.”
For now, the research gives scientists a clear target for designing the next generation of cholesterol drugs that could spare millions of people from unnecessary muscle pain.
Disclaimer: This article is for informational purposes only and is not intended as medical advice. If you are currently taking statins or experiencing muscle pain while on cholesterol-lowering medication, do not stop or change your treatment without consulting your healthcare provider. Statins have been proven to significantly reduce the risk of heart disease and stroke. Any medication changes should be made under medical supervision.
Paper Notes
Limitations
The study used very high concentrations of atorvastatin to get clear images of exactly where the drug binds, much higher than what circulates in patients’ blood. However, earlier studies measuring functional effects used concentrations that match real-world doses. The research used rabbit and pig muscle proteins rather than human ones, though the relevant parts of the proteins are nearly identical across species. The experiments left out ATP, a molecule that normally helps activate the protein, so how ATP and atorvastatin interact together in real muscle cells still needs investigation. Some evidence suggests atorvastatin might bind to a fourth location on the protein, but the images weren’t clear enough to be certain. Finally, while genetic differences predict that the heart and brain versions of the protein should resist atorvastatin, this wasn’t directly tested.
Funding and Disclosures
This research was supported by National Institutes of Health grant R01 HL170144 awarded to Héctor H. Valdivia and Filip Van Petegem. Cryo-electron microscopy data collection utilized the High Resolution Macromolecular Electron Microscopy facility at the University of British Columbia, funded by the Canadian Foundation for Innovation and British Columbia Knowledge Development Fund. The authors declare no competing interests. Commercial tissue sources included Pel-Freeze Biologicals (rabbit skeletal muscle) and Boyle Farms (pig skeletal muscle carrying the R615C mutation).
Publication Details
Molinarolo S, Valdivia CR, Valdivia HH, Van Petegem F. “Cryo-electron microscopy reveals sequential binding and activation of Ryanodine Receptors by statin triplets,” was published in unedited pre-print format on November 20, 2025 in Nature Communications. DOI:10.1038/s41467-025-66522-0. Authors are affiliated with the Department of Biochemistry and Molecular Biology, The Life Sciences Institute, University of British Columbia, Vancouver, British Columbia, Canada (Molinarolo, Van Petegem); and Department of Medicine, Division of Cardiovascular Medicine, and Cardiovascular Research Center, University of Wisconsin-Madison School of Medicine and Public Health, Madison, Wisconsin, USA (Valdivia CR, Valdivia HH).








I got the rare Charlie Horse in my heart from atorvastatin 10mg after having foot and leg cramps for several nights. All were extremely painful and lasted for several minutes. My heart felt bruised for two days and still have slight bruised feeling after discontinued use of six months with exertion. EKG was normal. I have Post Covid Syndrome of 7 years, and Type 2 Diabetes that I believe was caused by Covid plus many other symptoms. I don’t have a cholesterol issue and was taking the drug “just in case” according to PCP.
In order to eliminate statin based muscle pain, take at least 100mg of CoQ10 daily.
I read that when Lipitor was introduced, the manufacturer knew full well of this issue, but decided against adding CoQ10 to Lipitor due to cost measures. Pfizer first marketed Lipitor.
Hi Larry H,
A friend also suggested using CoQ10 when taking stains, but was unable to show me any Pharmalogical evidence it actually is a benefit to take.. This is not a comment of disbelief, however before ordering and taking CoQ10, I would like to see some peer reviews of this supplement and also who are the best suppliers of same. Again, please don’t take this as a criticism, it is only an attempt to gain more information.
I happen to be one of the unlucky few who have severe muscle pain from taking this statin for the past 10-yrs. Stopped taking it but still diagnosed with SINAM. Have you seen anything published that could reduce pain or muscle issues from this drug? Thank you!