
Photo by pasja1000 from Pixabay
ELCHE, Spain — Remember when everyone rushed to buy BPA-free water bottles and food containers? Well, those alternatives might not be as safe as we thought, according to new research. The study suggests that the chemical replacements for BPA behave almost identically to the controversial compound they were meant to improve upon.
For years, bisphenol A (BPA) has been the black sheep of the plastics family. This chemical, used in everything from water bottles to food can linings, gained notoriety for its ability to disrupt hormones in the body. As public concern grew, manufacturers began replacing BPA with alternative compounds, primarily bisphenol F (BPF) and bisphenol S (BPS), marketing their products as “BPA-free” and presumably safer.
However, José Villalaín from the Institute of Research, Development, and Innovation in Healthcare Biotechnology (IDiBE) at Universidad Miguel Hernández has uncovered some uncomfortable truths about these substitutes. Using sophisticated computer simulations, Villalaín examined how these chemicals interact with cell membranes – the protective barriers that surround all our cells. The findings? BPF and BPS behave remarkably similarly to BPA, suggesting they might affect our bodies in many of the same concerning ways.
“Given the environmental and health importance of these molecules, the use of these bisphenols should be discontinued due to the risks they may pose to human and animal health,” Villalaín says in a statement.
Think of cell membranes as the bouncers at a cellular nightclub. They control what gets in and out of the cell, maintaining order and protecting what’s inside. Villalaín discovered that all three bisphenols — BPA, BPF, and BPS — basically have the same VIP pass to get past these bouncers. They all hang out in similar areas of the membrane and interact with it in nearly identical ways.
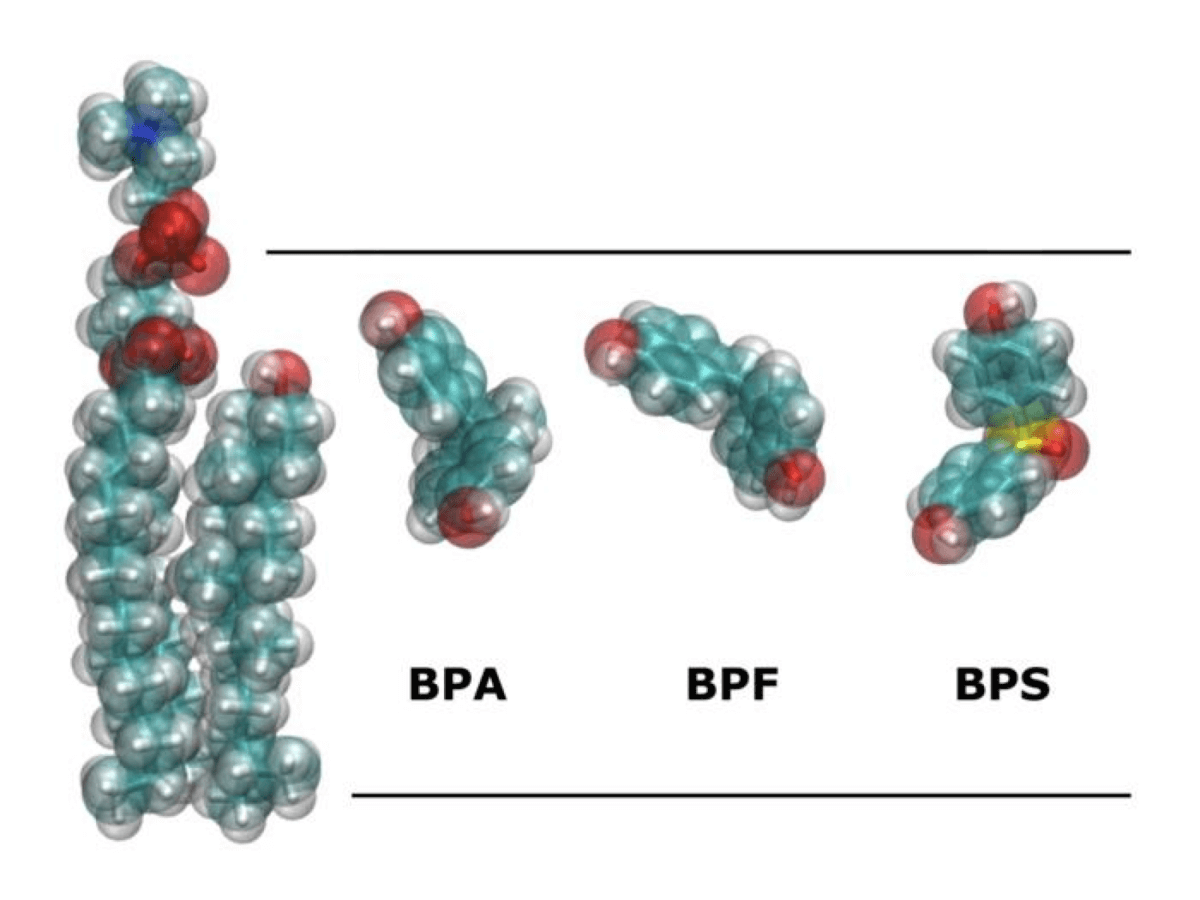
The study, published in the Journal of Xenobiotics, used complex molecular dynamics simulations – think of it as The Sims but for molecules – to watch how these chemicals behave when they encounter cell membranes. The researchers created various scenarios with different concentrations and arrangements of the bisphenols, running each simulation for an impressive 1,000 nanoseconds to see what would happen.
Strikingly, all three bisphenols showed a preference for positioning themselves just beneath the membrane’s surface, about six to eight angstroms deep (that’s roughly the width of a few atoms). They all had similar effects on membrane fluidity, basically making the membrane more fluid or “looser.” Perhaps most importantly, they all demonstrated the ability to form clusters or aggregates when present in higher concentrations, which could potentially affect cell function.
This clustering behavior is particularly concerning because while bisphenols tend to be excreted from our bodies within 24 to 48 hours, prolonged exposure means that over time, their levels can be relatively high in different organs, tissues, and cells. The continuous presence of these chemicals in our environment means they can maintain a consistent presence in our bodies despite our natural clearing mechanisms.
The research also revealed that these chemicals tend to avoid cholesterol in cell membranes, preferring to interact with other membrane components instead. This selective behavior could have implications for how these substances affect cellular processes, as cholesterol plays crucial roles in membrane organization and cell signaling.
What makes this study particularly compelling is its methodological approach. Rather than just looking at one scenario, Villalaín examined twelve different systems, varying the number and placement of bisphenol molecules. This comprehensive approach helped confirm that the behavior patterns observed weren’t just flukes but consistent characteristics of these chemicals.
The implications are clear: the “safer” alternatives to BPA might not be safer at all. In fact, they appear to be molecular copycats, mimicking the very behaviors that made BPA concerning in the first place. This finding adds to a growing body of evidence suggesting that BPF and BPS might be what scientists call “regrettable substitutions” – alternatives that turn out to be just as problematic as the original compound.
So, what’s a concerned consumer to do? While this study doesn’t provide direct evidence of harm in humans, it does raise important questions about the safety of BPA alternatives. It also highlights a broader issue in chemical safety: sometimes, replacing one concerning chemical with a structurally similar alternative isn’t the solution we think it is.
As Villalaín concludes in the paper, both BPF and BPS “are not safe alternatives to BPA” given their similar behaviors in cell membranes. It seems that, in this case, the apple doesn’t fall far from the chemical tree.
Paper Summary
Methodology
The study used computer simulations called molecular dynamics to observe how BPA, BPF, and BPS interact with cell membranes. The researchers created 12 different systems, each containing different amounts and arrangements of these chemicals, along with a model cell membrane that mimicked the complexity of real cell membranes. Each simulation ran for 1,000 nanoseconds, allowing researchers to track the movements and interactions of every molecule. They measured various parameters, including where the chemicals positioned themselves, how they affected membrane fluidity, and whether they formed clusters.
Key Results
The study found remarkable similarities in how BPA and its alternatives (BPF and BPS) interact with cell membranes. All three chemicals settled into the same location just below the membrane surface, made membranes more fluid, and formed clusters when present in higher concentrations. Notably, all three compounds avoided interacting with cholesterol while readily binding to other membrane components. These consistent patterns occurred across multiple experimental conditions, suggesting that BPA alternatives may affect cell membranes in ways virtually identical to BPA itself.
Study Limitations
The research relied solely on computer simulations rather than biological experiments, and the concentrations used may not reflect real-world exposure levels. Additionally, while sophisticated, the model membrane used was still simpler than actual cell membranes, and the study couldn’t directly measure health effects. These limitations suggest areas where future research could build upon these findings through laboratory and clinical studies.
Discussion & Takeaways
The key finding is that BPA alternatives (BPF and BPS) interact with cell membranes in ways virtually identical to BPA, suggesting they may not be safer alternatives. The study provides molecular-level evidence for why these alternatives might cause similar health concerns as BPA. The research also highlights how these chemicals can accumulate in cell membranes and form clusters, which could potentially disrupt cellular functions. This supports previous studies suggesting similar biological effects between BPA and its substitutes.
Funding & Disclosures
The research was partially funded by the “Programa de Ayudas para la Investigación del Envejecimiento del Centro Internacional para la Investigación del Envejecimiento de la Comunidad Valenciana, ICAR, Convocatoria 2023.” The author declared no conflicts of interest. The study used computing resources from SIPT, Universitas Miguel Hernández (UMH).







