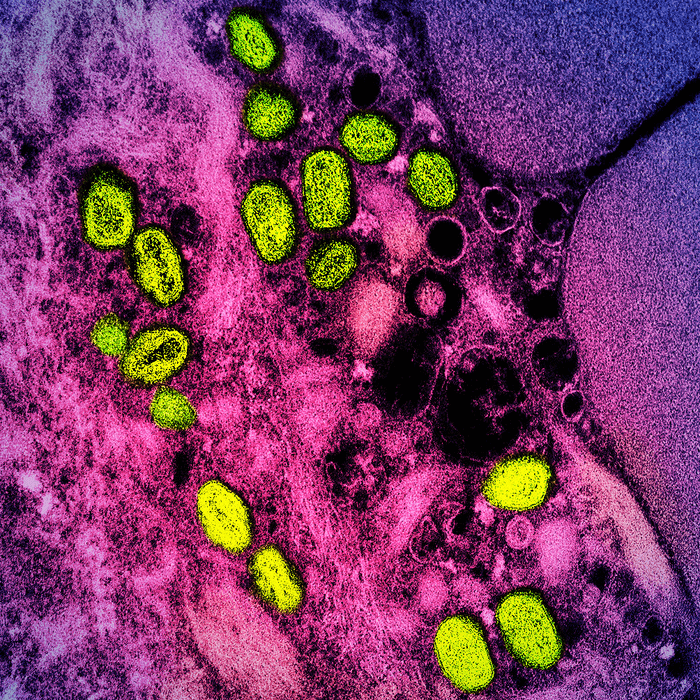
Colorized transmission electron micrograph of monkeypox particles (green) found within an infected cell (pink and purple), cultured in the laboratory. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. (CREDIT: NIAID)
ROCKVILLE, Md. — A pair of new clinical trials are underway which are testing the effectiveness of a new monkeypox drug and a new strategy for delivering the monkeypox vaccine.
The new Phase 3 clinical drug trial will evaluate tecovirimat, also known as TPOXX, among children and adults with a monkeypox infection in the United States. Investigators with the National Institutes of Health are enrolling 500 people nationwide to take part in the study.
Currently, tecovirimat is an FDA-approved drug used to treat smallpox. Researchers say the drug prevents the virus from spreading throughout the body by blocking virus particles from exiting human cells. TPOXX targets a protein which is present in both the variola virus, which causes smallpox, and the monkeypox virus.
At the moment, doctors can only gain access to supplies of tecovirimat for their patients with monkeypox through an expanded access or “compassionate use” request process.
“Monkeypox can be an extremely painful infection that lasts for weeks,” says NIAID Director Anthony Fauci in a media release. “We currently lack efficacy data that would help us understand how well this drug may mitigate painful monkeypox symptoms and prevent serious outcomes. This clinical trial was designed to answer those important questions.”
The Centers for Disease Control and Prevention has confirmed 21,504 cases of monkeypox across the United States as of Sept. 7. Prior to this global outbreak, monkeypox cases mostly took place in countries in central and west Africa, where the virus is endemic.
MORE: Monkeypox Update: Virus can now reportedly jump from humans to dogs
New vaccination method may increase supplies
While researchers test out a new drug to treat cases of monkeypox, another clinical trial will also begin examining a new delivery method for the monkeypox vaccine — which is in short supply.
The trial hopes to confirm that giving people an intradermal dose (between layers of the skin) of the JYNNEOS monkeypox vaccine is just as effective as current injection methods. This would allow doctors to use less of the vaccine in each dose — spreading out the supply.
Currently, health care professionals administer two subcutaneous (in the tissue beneath the skin) injections 28 days apart. However, the FDA recently authorized intradermal vaccines for adults. These shots carry one-fifth the standard dosage, which would allow doctors to vaccinate more people looking to avoid the virus.
“To halt the global outbreak of monkeypox and to help protect those at risk of infection, we need to ensure we have an adequate supply of monkeypox vaccine,” says Fauci, in a statement. “NIAID’s trial of JYNNEOS will provide important information on the immunogenicity, safety, and tolerability of alternative dosing approaches that would expand the current supply of vaccine.”
This trial will enroll more than 200 adults across eight U.S. research sites and is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID).
Those trial locations include:
- Saint Louis University in Missouri
- Baylor College of Medicine in Houston
- Brigham and Women’s Hospital in Boston
- The NIH Clinical Center in Bethesda, Maryland
- George Washington University in Washington, D.C.
- Vanderbilt University in Nashville, Tennessee
- The Hope Clinic at Emory University in Decatur, Georgia
- The University of California, San Diego







