
(Photo by Lisa Fotios from Pexels)
MADISON, Wis. — Vision is easy to take for granted, but just the thought of blindness is enough to terrify many people. Unfortunately, modern medicine offers frustratingly few treatment options once blindness sets in. However, researchers from the University of Wisconsin-Madison have made a stem cell breakthrough that could finally lead to a cure for degenerative eye disorders.
Scientists successfully demonstrated that retinal cells grown from stem cells are capable of “reaching out and connecting” with their neighboring cells. This cellular handshake, if you will, suggests that the cells are ready for clinical trials in humans dealing with vision loss, study authors adds.
Over a decade ago, researchers at UWM developed a way to grow organized clusters of cells (organoids), that resemble the retina — light-sensitive tissue at the back of the eye. Scientists then coaxed human skin cells reprogrammed to act as stem cells to develop into layers of numerous types of retinal cells that sense light, ultimately transmitting what we see to our brains.
“We wanted to use the cells from those organoids as replacement parts for the same types of cells that have been lost in the course of retinal diseases,” says David Gamm, the UW–Madison ophthalmology professor and director of the McPherson Eye Research Institute whose lab developed the organoids, in a university release. “But after being grown in a laboratory dish for months as compact clusters, the question remained — will the cells behave appropriately after we tease them apart? Because that is key to introducing them into a patient’s eye.”
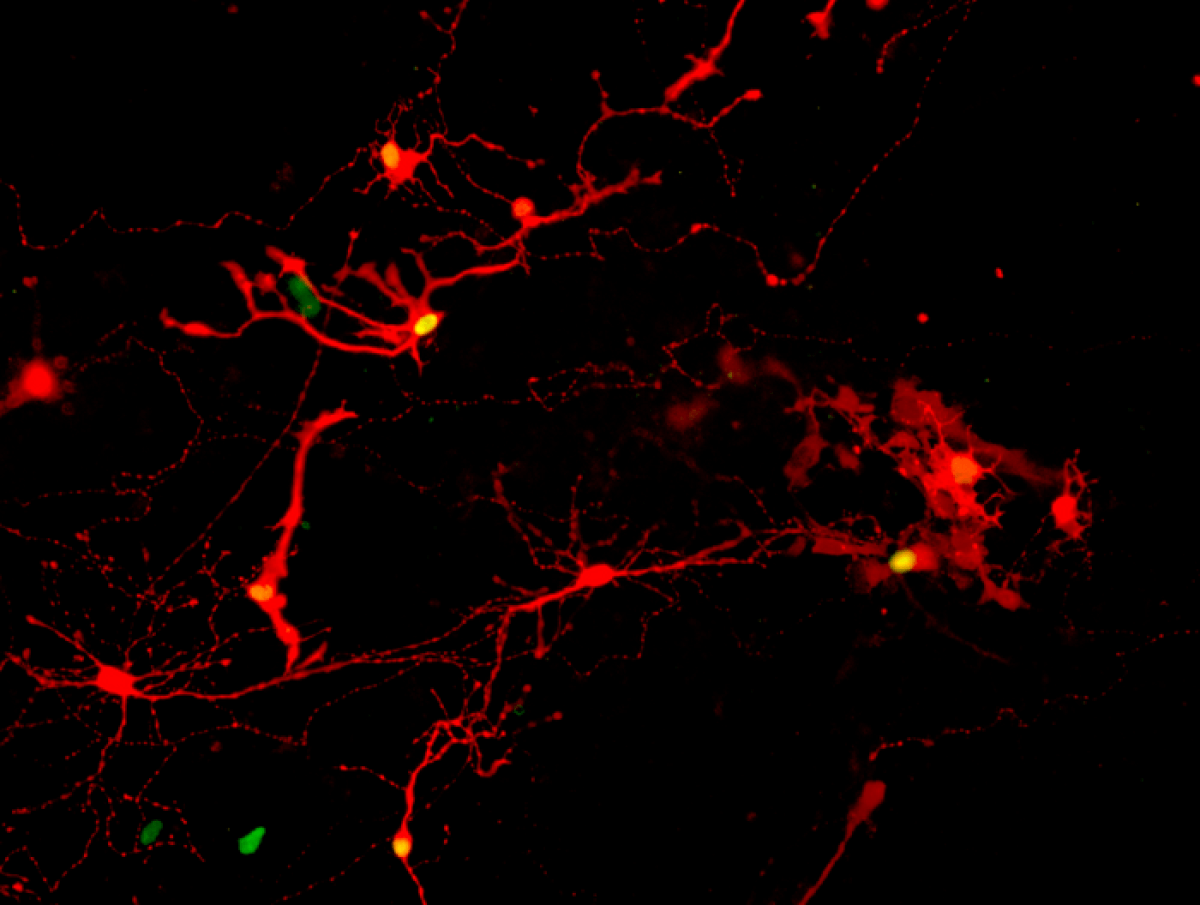
Synapses are the key to the puzzle
Last year, Prof. Gamm and additional UW–Madison collaborators published multiple studies showing that dish-grown retinal cells called photoreceptors respond like those in a healthy retina to different wavelengths and light intensities. Upon being separated from adjacent cells in their organoid, they can then reach out toward new neighbors with characteristic biological cords known as axons.
“The last piece of the puzzle was to see if these cords had the ability to plug into, or shake hands with, other retinal cell types in order to communicate,” adds Prof. Gamm.
Cells found in the retina and brain communicate across synapses, or tiny gaps at the tips of their cords. In order to confirm that their lab-grown retinal cells could replace diseased cells and carry sensory information just like healthy ones, study authors needed to show that they could make synapses in the first place.
Human trials are the next step
So, Xinyu Zhao, UW–Madison professor of neuroscience and study co-author, worked with the Gamm lab’s cells to study their ability to form synaptic connections. Zhao achieved this using a modified rabies virus which identifies pairs of cells that could form the means necessary to communicate.
Next, the research team, which included graduate students Allison Ludwig and Steven Mayerl, broke apart the retinal organoids into individual cells and waited a week to see if they would extend their axons and make new connections, exposed them to the virus, and then finally observed the results. They saw many retinal cells marked by a fluorescent color, indicating a rabies virus achieved infection across a synapse successfully formed between neighbors.
“We’ve been quilting this story together in the lab, one piece at a time, to build confidence that we’re headed in the right direction,” Prof. Gamm explains, who patented the organoids and co-founded Madison-based Opsis Therapeutics, which is adapting the technology to treat human eye disorders based on the UW–Madison discoveries. “It’s all leading, ultimately, to human clinical trials, which are the clear next step.”
After the team confirmed the presence of synaptic connections, they analyzed the cells involved and found that the most common retinal cell types forming synapses were photoreceptors (rods and cones). This is notable because photoreceptors are usually lost in diseases like retinitis pigmentosa, age-related macular degeneration, and even after certain eye injuries. The next most common cell type (ganglion cells) degenerate during optic nerve disorders like glaucoma.
“That was an important revelation for us,” Prof. Gamm concludes. “It really shows the potentially broad impact these retinal organoids could have.”
The study is published in Proceedings of the National Academy of Sciences.








When will human trials begin?
And if we can restore vision with stem cell, it shouldn’t take too long before we can regenerate hearts and brains with dementia. Human life will be much longer and healthier.
Do we know the source of the stem cells? Will they be from the patient so as to minimize rejection?