
Sticky traps are commonly used to get rid of fly infestations -- but are they in pain leading up to their deaths? (© Александр Гичко - stock.adobe.com)
In a nutshell
- Scientists proved for the first time that fruit flies can develop chronic pain-like states lasting weeks after injury, similar to human chronic pain conditions
- The research revealed that nerve injury kills crucial brain cells (GABA neurons) that normally prevent pain responses, directly causing the chronic pain state
- This discovery could lead to new treatments for human chronic pain by targeting cell death prevention or replacing lost inhibitory neurons
SYDNEY — Few people would hesitate to grab a newspaper and smash an annoying fly that’s been buzzing around the kitchen for hours. But if you’ve ever wondered whether bugs feel pain when you attempt to kill them, a 2019 study is the first to prove that not only do insects feel pain from an injury, but they suffer from chronic pain after recovering from one.
Researchers from the University of Sydney say that when a fruit fly loses a leg, something remarkable happens: the tiny insect enters a state of heightened vigilance that mirrors chronic pain conditions in humans. For weeks afterward, the injured fly will jump frantically at temperatures that wouldn’t normally trigger escape responses.
First published in Science Advances in July of 2019, this discovery could revolutionize treatments for the millions of Americans living with chronic pain conditions.
The researchers note that globally, chronic pain is estimated to cost trillions of dollars per year, and current therapies “do not adequately address pain for most patients.” With over 200,000 deaths from prescription opioid overdoses since 2000 (through the study period), finding new approaches has become urgent.
How Scientists Measured Pain-Like Responses in Fruit Flies
Led by Greg Neely, an associate professor at the university, scientists studied fruit flies after amputating one of their middle legs. While uninjured flies showed minimal response to moderately warm surfaces around 38°C (100.4°F), injured flies exhibited “thermal allodynia,” which are enhanced escape responses at temperatures that normally wouldn’t trigger such behavior.
The hypersensitive state didn’t appear immediately. Instead, it developed gradually over five to seven days after injury and persisted for at least three weeks, matching patterns seen in human chronic pain.
Study authors found that the pain-like behavior occurred even though the injured leg itself was no longer functional. When researchers stimulated the amputated leg directly, nothing happened. But when they stimulated the opposite, uninjured leg, the flies showed enhanced escape responses, jumping faster and for longer durations than healthy flies.
This pattern shows that chronic pain-like states involve changes in the central nervous system rather than just the injury site, matching what scientists observe in human chronic pain patients.
“People don’t really think of insects as feeling any kind of pain,” Neely explains in a statement. “But it’s already been shown in lots of different invertebrate animals that they can sense and avoid dangerous stimuli that we perceive as painful. In non-humans, we call this sense ‘nociception’, the sense that detects potentially harmful stimuli like heat, cold, or physical injury, but for simplicity we can refer to what insects experience as ‘pain’.
“So we knew that insects could sense ‘pain’,” he continues, “but what we didn’t know is that an injury could lead to long lasting hypersensitivity to normally non-painful stimuli in a similar way to human patients’ experiences.”

The Biological Domino Effect Behind Chronic Pain In Insects
Digging deeper into the biological mechanisms, researchers discovered that injury triggers a cascade of cellular destruction in the flies’ nervous systems. Specifically, nerve damage causes the death of cells that normally keep pain responses in check—neurons that use a chemical messenger called GABA to dampen nervous system activity.
When scientists prevented these crucial cells from dying using genetic techniques, injured flies never developed chronic pain-like behaviors. Conversely, artificially blocking these inhibitory cells was enough to create chronic pain-like behavior in healthy flies. This represents some of the strongest evidence to date that the death of these dampening neurons directly causes chronic pain.
The molecular pathway reads like a cellular domino effect: injury activates sensory neurons that release acetylcholine, which overstimulates the GABA neurons through specific receptors. This overstimulation triggers a protein called Twist, which ultimately leads to programmed cell death.
Each step in this pathway proved essential. When researchers blocked any single component — whether the receptors, the Twist protein, or the final cell death machinery — chronic pain failed to develop.
Ancient Pain Systems May Hold Modern Solutions
The research suggests this pain response might actually be beneficial from an evolutionary perspective. Injured animals in the wild face increased predation risk, and a heightened state of vigilance could improve survival chances after serious injury.
Pain systems are remarkably conserved across species; the same temperature-sensitive channels that detect harmful heat in flies also function in human pain perception. Many genes involved in fly pain responses have direct counterparts in mammals.
Human chronic pain patients often show reduced GABA activity in their spinal cords, matching what researchers observed in the fly nervous system. Some experimental treatments for human chronic pain, including transplanting GABA-producing cells into the spinal cord, have shown promise in reversing pain symptoms.
However, definitive proof that inhibitory cell death causes chronic pain has been nearly impossible to obtain in mammals due to ethical and technical constraints. The fly model offers a genetically tractable system where researchers can precisely manipulate specific cell types to test causation rather than just correlation.
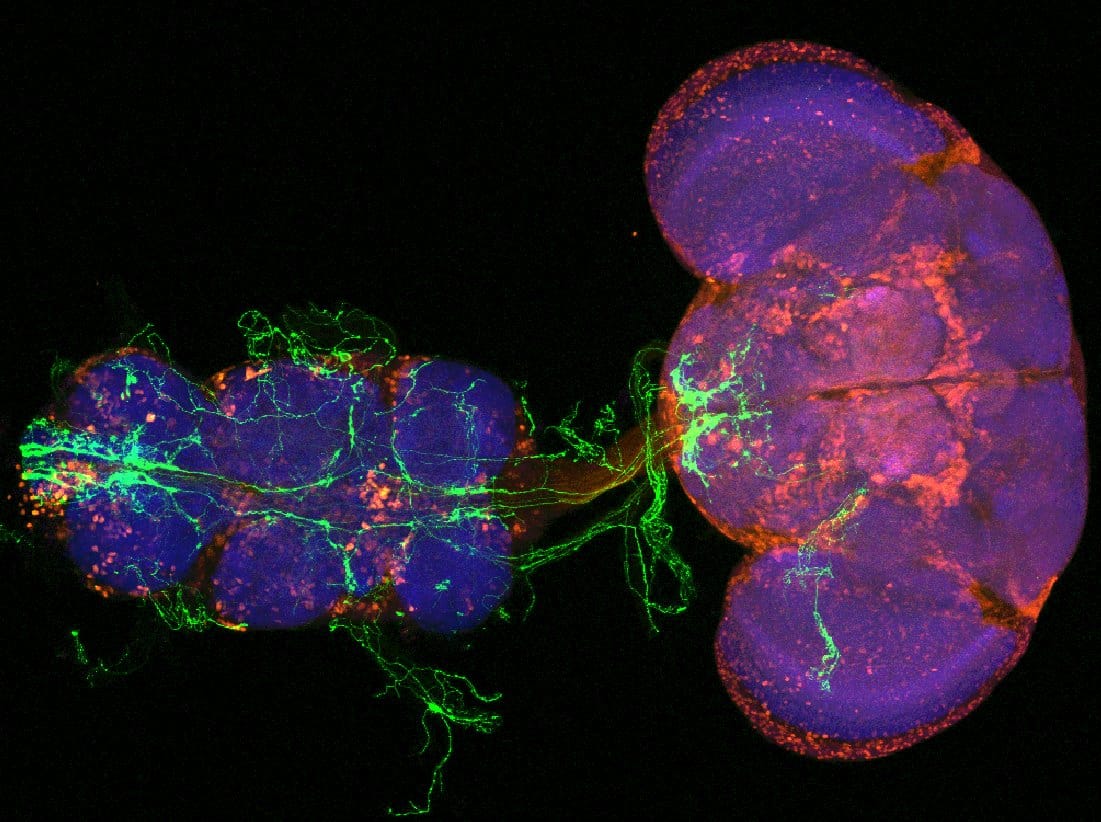
What Humans Can Learn From Studying Whether Bugs Can Feel Pain
The research team tested their flies using a custom-built “hot plate” apparatus, where groups of 10 flies were exposed to gradually increasing temperatures while cameras recorded their jumping behavior. Across multiple experiments involving hundreds of flies, the results were consistent: injury led to enhanced escape responses at normally harmless temperatures.
While fruit flies might seem like unlikely candidates for pain research, they share fundamental neural architecture with humans. Both species use similar molecules to detect harmful stimuli, process sensory information, and generate protective responses.
Current chronic pain treatments often fail because they target symptoms rather than underlying causes. Learning that inhibitory cell death drives chronic pain-like states could point toward new therapeutic approaches focused on protecting these crucial neurons or replacing their function.
Some existing drugs that prevent cell death or enhance GABA signaling might warrant renewed investigation as chronic pain treatments. The research also identified several molecular targets that hadn’t previously been considered for pain therapy.
Moving forward, researchers can use the fly model to rapidly screen potential therapeutic compounds, test combination treatments, and learn how genetic variations might influence chronic pain susceptibility.
“Importantly now we know the critical step causing neuropathic ‘pain’ in flies, mice and probably humans, is the loss of the pain brakes in the central nervous system,” says Neely. “We are focused on making new stem cell therapies or drugs that target the underlying cause and stop pain for good.”
So yes, as it turns out, insects may not only feel pain, but they can battle chronic pain just like us. This study shows that it’s not just a human problem or even a vertebrate problem, but a fundamental feature of how nervous systems respond to injury across hundreds of millions of years of evolution. Cracking this ancient machinery could finally provide the keys to treating one of humanity’s most persistent sources of suffering.
Paper Summary
Methodology
Researchers used adult fruit flies (Drosophila melanogaster) and developed a thermal nociception assay system to measure pain-like responses. They amputated the right middle leg of flies and tested their escape responses to different temperatures (25°C to 42°C) over time periods ranging from 1 to 21 days after injury. The experiments used groups of 10 flies tested in behavioral chambers with variable heat elements, and researchers recorded jumping behavior using video cameras. They also performed electrophysiological recordings from escape muscles and used genetic techniques to manipulate specific types of neurons. Immunohistochemistry was used to examine changes in brain and nervous system tissues.
Results
Injured flies developed thermal allodynia—enhanced escape responses to normally harmless temperatures (38°C)—starting 5-7 days after injury and persisting for at least 21 days. This response required specific temperature-sensitive channels (TrpA1 and painless) and sensory neurons (ppk+ neurons). The researchers discovered that injury causes death of inhibitory GABA neurons in the central nervous system through a pathway involving acetylcholine signaling, nAChRα1 receptors, Twist protein, and caspase-dependent cell death. Preventing GABA cell death blocked chronic pain development, while artificially reducing GABA signaling was sufficient to cause pain-like behavior without injury.
Limitations
The study was conducted only in fruit flies, so the relevance to human chronic pain requires further validation. The researchers focused on thermal sensitivity rather than other types of pain, and the injury model (leg amputation) represents just one type of nerve damage. While the molecular pathways appear conserved across species, the subjective experience of pain in insects remains debatable. The study also examined only male flies and didn’t investigate sex differences in pain responses.
Funding and Disclosures
This work was supported by multiple grants from the Australian National Health and Medical Research Council (NHMRC), including project grants APP1026310, APP1029672, APP1028887, APP1046090, APP1042416, and APP1086851. The lead researcher G.G. Neely was supported by an NHMRC career development fellowship. Additional funding came from J. Chong and A. Chong. The authors declared no competing interests.
Publication Information
The study was published in Science Advances on July 10, 2019, by Thang M. Khuong, Qiao-Ping Wang, John Manion, Lisa J. Oyston, Man-Tat Lau, Harry Towler, Yong Qi Lin, and G. Gregory Neely from the University of Sydney and Sun Yat-sen University. The paper is titled “Nerve injury drives a heightened state of vigilance and neuropathic sensitization in Drosophila” (doi: 10.1126/sciadv.aaw4099).
This is an updated version of an article that was first published July 13, 2019.








Nociception is not the same thing as pain. Just because an animal tries to get away from potentially harmful stimuli, like heat, cold, or pressure, doesn’t necessarily mean that the animal feels what we know as pain from those stimuli. This seems a little misleading, although the “pain brakes” idea is intriguing. Just saying…
Nociception and pain are not the same thing as is implied here.
If you’ve ever almost smashed a fly and watched it buzz in circles, you watched it experience agony. It’s easy to make the connection if you have been In agony,flailing around while the EMT implores you to be still, as you, through gritted teeth,grunt out, It Hurts too bad. 45 MPH into the side of a car on a motorcycle is a fair comparison.
Yes.. very sensitive creatures.
you, my man are a informed person who knows things and can sense horrible people at the slightest sense
How is this transmitted in the insect? Do they have neurotransmitters, chemicals, and electrical transmission along the cord?
I love the screaming of horror i give with my destruction of yellow jacket ground nests, nasty buggers deserve it.
you are the scum of the earth and need to be eradicated
What in God’s name are you talking about?!
I don’t particularly like yellow jackets either. I stepped in a nest as a child and was stung over 70 times. However, it doesn’t give us the right to cause any living creature unnecessary pain. If you must kill them, fine. But I cannot understand your glee in doing so. They are simply insects without the mental capacity to want to harm us.
Take it down a few notches maybe? And unlock your caps lock, Sister Mary Yellinatme.
I think it is just like when someone hurt you badly in the past and you become “hypersensitive” in this area and “try to protect yourself for the rest of your live”…
If it wasn’t obvious, the study just gives us more insight into the neuropathic responses of insects and that this feature of the responses is similar to what we see in humans. We still don’t know what it’s actually like to be a fly and be injured. Hence, “pain” in quotes.