Credit: PeopleImages on Shutterstock
The eyes are often said to be a window to the soul. This study suggests they may offer a better view of the mind.
In A Nutshell
- Researchers found that twisted retinal blood vessels and arterial-venous crossing patterns appeared as early as 6 months in mice with the MTHFR677C>T variant, mirroring brain damage happening simultaneously
- The genetic variant, carried by up to 40% of people worldwide, causes toxic homocysteine buildup that damages blood vessels in both the eyes and brain through the same molecular pathways
- Blood vessel abnormalities showed up before any measurable loss of retinal function, suggesting a potential window for early intervention
- While the research is still in mice, it raises the possibility that routine eye exams could one day help identify people at risk for vascular dementia before memory problems begin
Your next eye exam could reveal more than whether you need new glasses. Scientists have discovered that changes in tiny blood vessels at the back of the eye can signal brain damage in mice with a genetic variant linked to dementia. This suggests routine vision checkups may one day serve as an early warning system for cognitive decline in people.
Researchers at The Jackson Laboratory found that retinal blood vessels mirror what’s happening in the brain when cerebrovascular disease develops. The team studied mice carrying MTHFR677C>T, a common genetic variant found in 20% to 40% of people worldwide that has been associated with increased risk for Alzheimer’s disease and vascular dementia.
The researchers wanted to determine if changes in the retina reflect similar changes happening in brain blood vessels. According to their study published in Alzheimer’s & Dementia, the answer is yes.
What Researchers Found in Mouse Eyes
Using standard ophthalmology equipment similar to what eye doctors use in clinics, researchers photographed retinal blood vessels in mice at 6 and 12 months of age. Female mice with the variant showed the most pronounced changes.
The researchers observed twisted vessels and instances where arteries pressed down on veins, narrowing blood flow. Both abnormalities are warning signs doctors already look for when assessing stroke and cardiovascular disease risk in human patients. Arteries in affected mice became narrower while veins grew wider, suggesting the vascular system was struggling with altered blood pressure.
These specific abnormalities appeared at 6 months, while broader loss of small blood vessels didn’t show up until 12 months. The timing matters because twisted vessels and arterial-venous crossings might serve as earlier markers than overall vessel density changes.
Tests of retinal function showed no differences between normal and variant-carrying mice at 6 months, and retinal thickness remained normal. This means blood vessel abnormalities appeared before measurable functional changes, potentially offering a window for intervention.
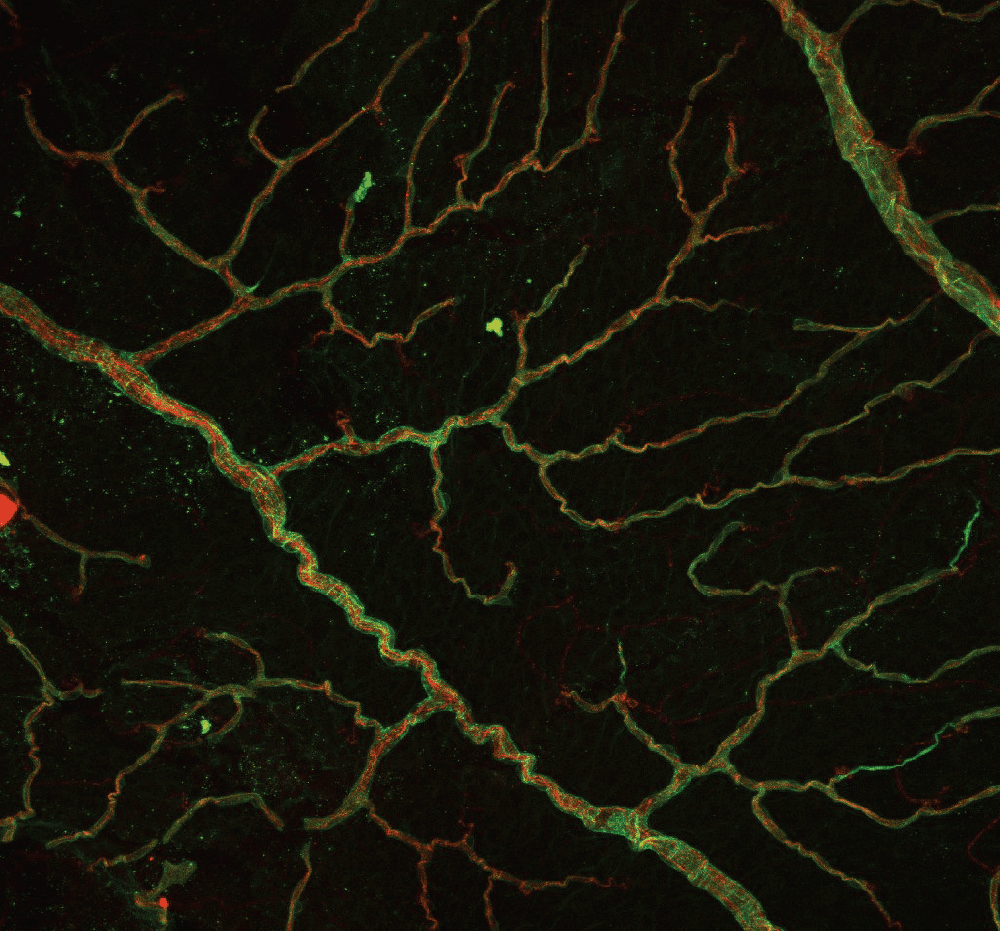
How a Common Gene Variant Damages Blood Vessels
The MTHFR gene produces an enzyme for processing folate (vitamin B9) and regulating homocysteine, an amino acid that becomes toxic at high levels. When the enzyme works poorly due to this genetic variant, homocysteine accumulates and damages blood vessels throughout the body.
People carrying one or two copies of the variant may have chronically elevated homocysteine levels, especially if their diet lacks sufficient folate. Elevated homocysteine has been associated with increased risk for cardiovascular disease, stroke, and cognitive decline in human studies, though this mouse research doesn’t directly test those outcomes.
The research team found the MTHFR gene active throughout the retina and brain, particularly in blood vessel cells. When they analyzed tissue samples, they discovered the same molecular stress signals in both locations, suggesting the variant creates parallel damage.
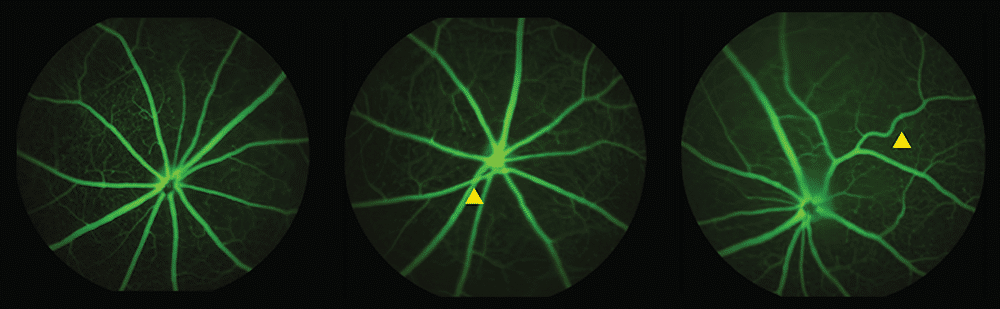
From Mice to Potential Human Applications
Retinal imaging is non-invasive, relatively inexpensive, and already part of routine eye care. If future research confirms that retinal vascular changes can predict cerebrovascular disease in humans, ophthalmologists might be able to identify at-risk patients earlier.
Previous studies have linked retinal abnormalities to Alzheimer’s risk in people, and doctors already use retinal vessel patterns to assess cardiovascular disease. What makes this mouse study different is demonstrating that a specific genetic variant causes matching dysfunction in both the eye and brain.
The researchers note several limitations. The mice were maintained on standard laboratory chow with folic acid levels that may not fully compensate for the enzyme deficiency. Additionally, the variant alone doesn’t cause dementia in these animals but rather creates vascular vulnerability that could worsen with other risk factors like aging or high blood pressure.
The team plans to track both brain and retinal blood vessels in the same animals over time to directly correlate changes as disease progresses. They also want to introduce additional Alzheimer’s-related genetic changes to better model human disease.
While this research is still in early stages and conducted in mice, it establishes biological plausibility for using eye exams to assess brain vascular health.
Disclaimer: This article discusses early-stage research in mice and is not medical advice. People concerned about dementia risk or carrying the MTHFR variant should consult their healthcare provider about appropriate screening and management.
Paper Summary
Limitations
The research used mice maintained on standard laboratory diet containing folic acid at approximately one-fourth the CDC-recommended human daily allowance. This may not fully compensate for MTHFR enzyme deficiency. The study examined only two time points and used bulk tissue analysis rather than isolating specific cell types. Sample sizes ranged from 6 to 20 animals per group depending on the experiment.
Funding and Disclosures
This work was funded by an anonymous donation and the Diana Davis Spencer Foundation. Gareth Howell holds the Diana Davis Spencer Foundation Chair for Glaucoma Research. The National Institute of Neurological Disorders and Stroke provided additional support (Grant NS139948). The authors declare no conflicts of interest.
Publication Details
Reagan AM, MacLean M, Cossette TL, Howell GR. “Retinal vascular dysfunction in the Mthfr677C>T mouse model of cerebrovascular disease.” Alzheimer’s & Dementia. 2025;21:e70501. DOI: 10.1002/alz.70501. The research was conducted at The Jackson Laboratory, Bar Harbor, Maine, with additional affiliations at Tufts University School of Medicine and the University of Maine Graduate School of Biomedical Sciences and Engineering.







