
Researchers found that gum disease can lead to more of the bacteria that cause atrial fibrillation getting into the heart. (New Africa/Shutterstock)
In a nutshell
- Researchers found that Porphyromonas gingivalis, a common gum disease bacterium, can travel from infected gums to the heart, where it may contribute to atrial fibrillation by promoting tissue scarring.
- The severity of gum inflammation, not just gum pocket size, was strongly associated with the amount of bacterial DNA found in heart tissue and the degree of atrial fibrosis in both mice and human patients.
- The study suggests that treating periodontal disease could potentially reduce the risk or progression of atrial fibrillation, highlighting oral hygiene as a possible factor in heart rhythm management.
HIROSHIMA, Japan — Your dentist’s warnings about flossing aren’t just about preventing bad breath and cavities. New research from Japan reveals a disturbing connection between gum disease and one of America’s most common heart conditions.
Scientists have discovered that bacteria from infected gums can travel through your bloodstream directly to your heart, potentially triggering atrial fibrillation, an irregular heartbeat that affects millions of Americans and significantly increases stroke risk.
The study, published in the journal Circulation, found that a common oral bacterium called Porphyromonas gingivalis (P. gingivalis) can escape from inflamed gum tissue, enter the bloodstream, and make its way to the left atrium of the heart. Once there, these bacteria appear to promote scarring of heart tissue, creating favorable conditions for abnormal heart rhythms to develop.
The research shows that patients with severe gum disease have higher levels of these bacteria in their heart tissue, which appears to worsen heart scarring and lead to poorer outcomes for those with atrial fibrillation.
This discovery provides a potential explanation for something cardiologists and dentists have observed for years: people with gum disease seem to have higher rates of heart problems, but the exact reason linking the two conditions remained unclear until now.
The research team, led by scientists from Hiroshima University in Japan, conducted experiments on both mice and humans. They deliberately infected mice with P. gingivalis by placing the bacteria in their molars and sealing them with a resin cap. This created a localized infection mimicking human periodontitis (advanced gum disease).
Using sophisticated tracking techniques, the researchers watched as the bacteria traveled from the dental infection through the bloodstream and into the heart. The infected mice developed increased heart tissue scarring and were more likely to develop atrial fibrillation when their hearts were electrically stimulated compared to healthy mice.
The longer the infection persisted, the worse the outcomes became. Mice examined 18 weeks after infection showed a significantly higher rate of atrial fibrillation (30%) compared to uninfected mice (5%).
The researchers also examined tissue samples from the left atrial appendages of 68 human patients who were undergoing cardiac surgery for atrial fibrillation. These patients also received dental examinations before surgery.
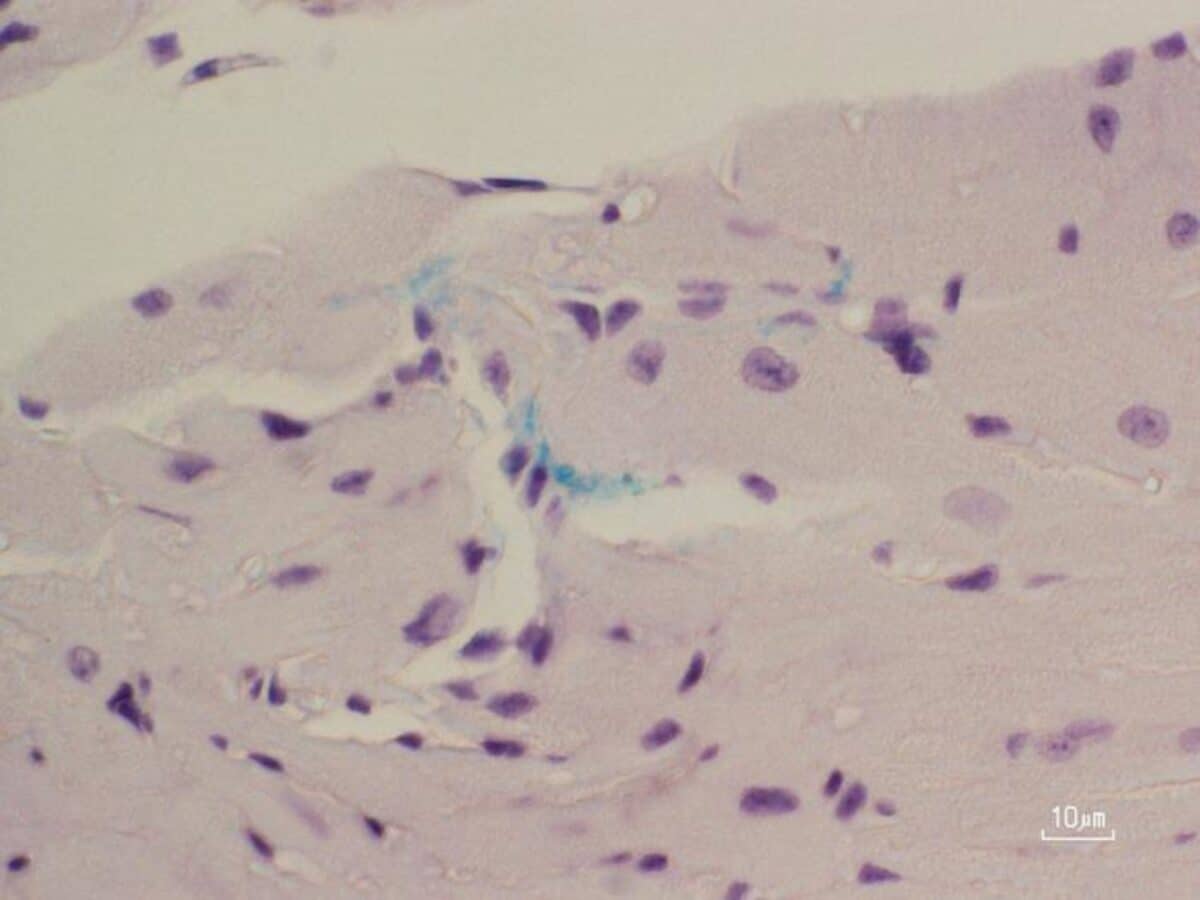
Patients with worse periodontal disease harbored more P. gingivalis bacteria in their heart tissue. The amount of bacteria found in the heart directly correlated with the degree of heart tissue scarring.
Researchers measured the severity of gum disease using two metrics: the periodontal epithelial surface area (PESA), which represents the total surface area of infected gum pockets, and the periodontal inflamed surface area (PISA), which indicates the amount of actively inflamed gum tissue.
PISA values showed an especially strong correlation with the amount of P. gingivalis found in heart tissue and the degree of heart scarring. This suggests that active inflammation in the gums, rather than just the presence of gum pockets, is what creates the opportunity for bacteria to escape into the bloodstream.
This bacterial invasion appears to be related to specific inflammatory pathways. The researchers found that P. gingivalis infection was associated with increased levels of several inflammatory molecules, particularly galectin-3 and transforming growth factor beta 1 (TGF-β1), which are known to promote tissue scarring.
This microscopic chain of events, from gum infection to bacterial travel through the bloodstream to heart inflammation and scarring, provides a plausible explanation for why people with gum disease have a higher risk of developing atrial fibrillation.
This discovery could change the lives of the approximately 12 million Americans currently living with atrial fibrillation. Good oral hygiene and regular dental care might be more than just cosmetic concerns; they could be essential components of heart health management.
If P. gingivalis is indeed contributing to the development or progression of the condition, therapies targeting this specific bacterium might help some patients. The authors suggest that not only gum disease but the bacterium itself could serve as a treatment target for atrial fibrillation.
While the research is still in its early stages, it adds to a growing body of evidence that oral and cardiovascular health are intimately connected. Similar associations have been found between gum disease and other conditions like arterial plaque buildup, diabetes, and even Alzheimer’s disease.
Taking care of your gums isn’t just about preserving your smile; it could also protect your heart. We might need to look no further than our toothbrushes and dental floss for our heart’s first line of defense.
Paper Summary
Methodology
Researchers conducted experiments on both mice and humans to investigate the relationship between periodontitis, bacterial translocation, and atrial fibrillation. For the animal study, they created a mouse model by inoculating P. gingivalis (a common periodontal pathogen) into the pulp of mice molars and sealing it with resin caps. They then examined these mice after 12 and 18 weeks, using immunohistochemistry to track the bacteria’s path from the dental infection to the heart and measuring atrial fibrosis and AF inducibility. For the human study, they collected left atrial appendage specimens from 68 patients with AF undergoing cardiac surgery. Before surgery, patients underwent periodontal examinations to measure the severity of periodontitis using PESA (periodontal epithelial surface area) and PISA (periodontal inflamed surface area) metrics. The researchers then quantified P. gingivalis in the heart tissue samples and measured atrial fibrosis.
Results
The mouse experiments showed P. gingivalis successfully translocated from dental infections to the left atrium via the bloodstream. Infected mice developed significantly more atrial fibrosis (21.9% vs. 16.3% in controls) and had higher AF inducibility (30% vs. 5% in controls after 18 weeks). In human patients, the amount of P. gingivalis found in heart tissue positively correlated with periodontal disease severity, particularly PISA values. P. gingivalis levels also correlated with the degree of atrial fibrosis. Molecular analysis showed upregulation of inflammatory markers like galectin-3 and TGF-β1 in infected tissues, suggesting a potential mechanism linking bacterial infection to fibrosis development. The relationship between P. gingivalis and atrial fibrosis remained significant even after adjusting for other clinical factors.
Limitations
The study acknowledged several limitations. The animal model may not fully replicate human periodontal disease. The human heart samples came exclusively from patients with existing cardiac conditions requiring surgery, not from healthy individuals. The cross-sectional design with a relatively small sample size (68 patients) limits causal inference. The researchers also noted potential selection bias, especially in patients referred for certain procedures. Additionally, bacterial quantification was performed on single sections of heart tissue, which might not represent the entire atrial bacterial distribution.
Funding and Disclosures
The study was supported by the Japan Society for the Promotion of Science (Tokyo, Japan) through a Grant-in-Aid for Research Activity Start-Up (21K20924) and a Grant-in-Aid for Scientific Research (B; 21H03112). The authors declared no conflicts of interest related to the research.
Publication Information
The study titled “Atrial Translocation of Porphyromonas gingivalis Exacerbates Atrial Fibrosis and Atrial Fibrillation” was published in the journal Circulation in 2025. The research was led by Dr. Shunsuke Miyauchi and colleagues from various departments at Hiroshima University in Japan, including the Departments of Cardiovascular Medicine, Bacteriology, Oral and Maxillofacial Pathobiology, and General Dentistry.








I’m of an age when the professional telling me about my “deep pockets” is not my accountant, but my dentist.