MRI of Alzheimer's dementia patient's brain. (Credit: © Atthapon Raksthaput | Dreamstime.com)
Another piece of the dementia puzzle may finally have been found.
In A Nutshell
- Scientists discovered ultrathin tubes (hundreds of nanometers wide) connecting brain cells that transport proteins and calcium signals alongside traditional synapses
- These “dendritic nanotubes” actively move Alzheimer’s proteins between neurons at speeds consistent with motor protein transport, potentially explaining how the disease spreads
- The network changes 3 months before plaques form in mouse models, offering a possible early detection target that precedes current diagnostic markers
- Cells with fewer nanotube connections become toxic dumping grounds for amyloid proteins while well-connected cells share the burden, creating a vicious cycle of accumulation and isolation
Scientists studying Alzheimer’s have long wondered how exactly toxic proteins spread from neuron to neuron, leaving some cells devastated while their neighbors remain unscathed. Now, researchers have discovered a hidden network of ultrathin tubes connecting brain cells. The structures, called dendritic nanotubes, appear to change months before telltale amyloid plaques form, which might eventually help identify the disease earlier.
For over a century, researchers believed neurons communicated primarily through synapses, microscopic junctions where chemical signals jump between cells. A team at Johns Hopkins University has now identified an entirely separate communication network operating alongside them. These nanoscale tunnels, hundreds of nanometers wide and extending for micrometers, create direct bridges between the branching dendrites of neighboring neurons.
Always There, Finally Seen
The discover, described in a paper published in Science, emerged when Hyung-Bae Kwon and colleagues reexamined high-resolution electron microscopy images of mouse and human brain tissue. The images had been sitting in public databases, acquired for other research purposes, waiting for someone to notice what was hiding in plain sight.
Kwon’s team found thin protrusions extending from dendrites that lacked the telltale features of synapses. Instead, these structures formed closed-ended contacts with neighboring dendrites, primarily at the tips of the extensions. In human brain tissue, longer protrusions were particularly likely to establish these contacts.
To study the structures in living tissue, the researchers used mice genetically engineered to express fluorescent protein in scattered neurons. Advanced microscopy revealed bridge-like extensions rich in actin, the protein that forms cellular scaffolding. Unlike typical neuronal branches, these lacked another structural protein called tubulin.
Time-lapse imaging over 66 hours showed these tunnels form and disappear dynamically, with most having shorter lifespans than stable branches. When researchers blocked actin assembly with a chemical inhibitor, both the formation and length of these structures plummeted, confirming they depend on active cellular processes requiring energy.
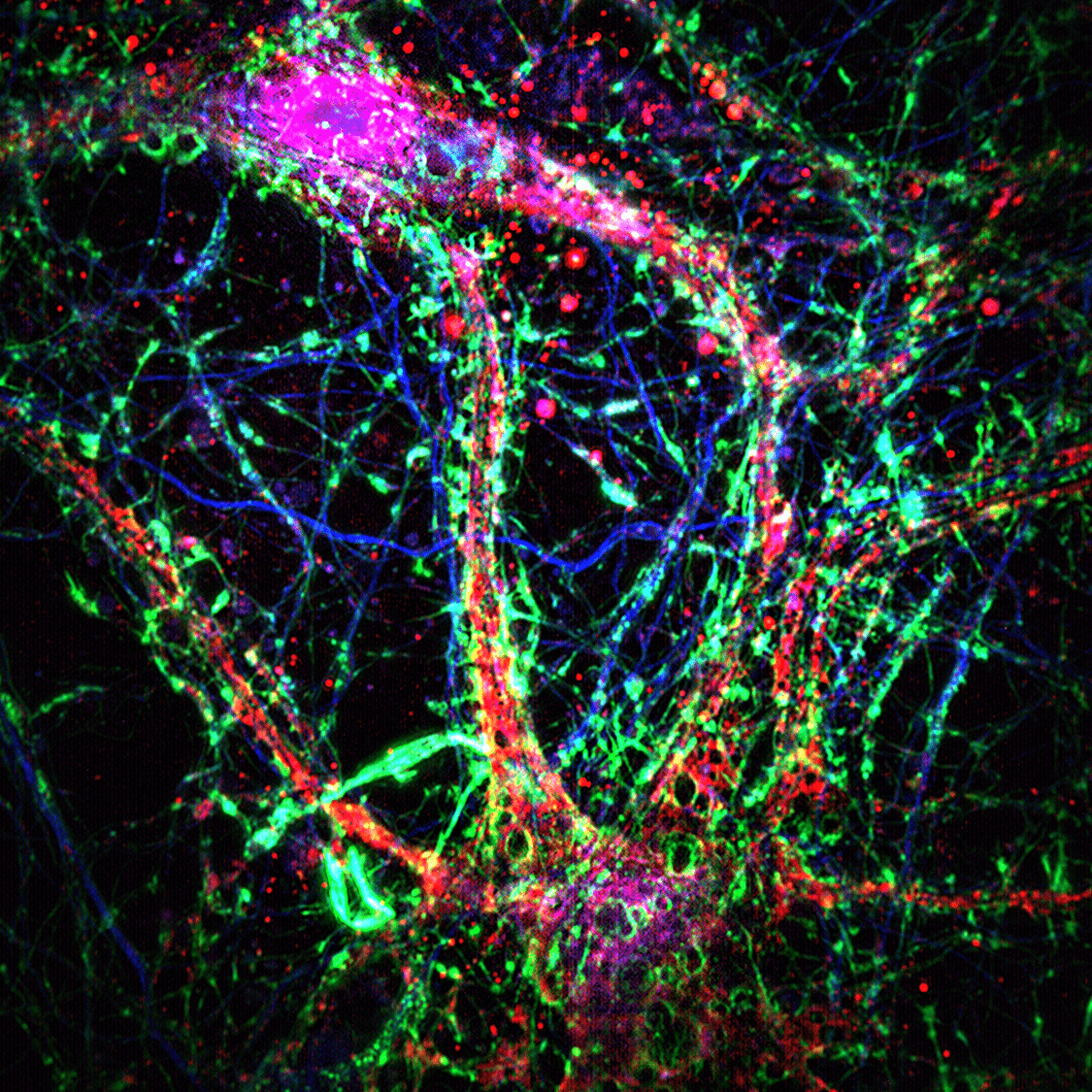
A Highway for Toxic Proteins
The critical question remained: do these nanotubes actually transport molecules between cells? The researchers focused on amyloid-beta, the toxic peptide that accumulates in Alzheimer’s brains.
In a clever experiment, they injected a single neuron with trackable amyloid-beta proteins. Over 30 minutes, neighboring neurons began showing amyloid signals inside them. Some recipient cells sat more than 200 micrometers away from the injection site. Control experiments where the solution simply leaked outside the cell showed virtually no transfer, confirming the nanotubes were creating direct cell-to-cell pathways.
When researchers disrupted the nanotube network, amyloid-beta couldn’t spread. The peptides didn’t drift passively through the tubes either. Tracking experiments revealed they moved rapidly along neuronal branches at speeds around 4 to 5 micrometers per second, consistent with motor proteins actively hauling cargo. Movement along the nanotubes themselves was slower but still directed.
Early Alzheimer’s Disease Warning Signs
Given this capacity to transport amyloid-beta, the team examined whether the nanotube network changes as Alzheimer’s develops. They studied mice engineered to develop the disease, examining brains at three and six months of age.
At three months, before amyloid plaques had formed, neurons showed increased formation of these nanotubes and much greater variability between cells. By six months, when plaques were abundant throughout the brain, the elevated formation had subsided, but the cell-to-cell variability remained high. Neurons with relatively high amounts of toxic amyloid showed reduced ability to form new nanotubes.
This timeline matters: the network changes appear months before the plaque buildup characteristic of Alzheimer’s disease.
Why Some Cells Suffer More
To understand how nanotube networks affect disease progression across many neurons, the researchers built computer simulations modeling amyloid dynamics in 1,668 interconnected cells. The simulations started all neurons with modest amyloid levels, mimicking early disease stages.
The results revealed a troubling pattern. Only certain cells reached toxic concentrations, and these were consistently the neurons with the fewest nanotube connections. Well-connected neurons could efficiently export amyloid to neighbors, but poorly connected neurons became collection points for the toxic peptide. As amyloid accumulated in these vulnerable cells, their ability to form new nanotubes declined further, creating a vicious cycle.
Experiments with cultured neurons confirmed this model. Exposing cells to low amyloid concentrations for 24 hours increased nanotube formation without causing obvious accumulation inside cells. At higher concentrations that did cause buildup, nanotube formation decreased.
The Therapeutic Challenge
The research suggests Alzheimer’s progression is driven partly by an altered cellular network. In early stages, increased nanotube formation might allow neurons to share the burden of clearing toxic proteins. But as some cells become overloaded, they lose the ability to participate in this sharing network, becoming isolated repositories that eventually die.
For researchers seeking interventions, this creates a genuine dilemma. Boosting nanotube formation might help some neurons shed toxic proteins, but it could also accelerate disease spread to vulnerable neighbors with fewer connections. Blocking the tubes might protect individual cells from receiving toxic cargo, but it could prevent the network-level sharing that keeps the system healthy overall.
The stakes extend beyond Alzheimer’s. These dendritic nanotubes appear to operate as a parallel communication system, transmitting both normal signals like calcium and pathological cargo like toxic proteins. Other neurodegenerative diseases involving protein spread between cells, including Parkinson’s and Huntington’s disease, may exploit similar pathways.
What Comes Next
The immediate priorities are mapping how these nanotubes are distributed across different brain regions and determining whether they can be pharmacologically manipulated without disrupting normal brain function.
The hidden highways connecting neurons now offer a fundamentally new way to understand how Alzheimer’s disease spreads through the brain. For patients and families affected by this devastating illness, that understanding may eventually translate into interventions that target the disease before it destroys cognition and memory.
The structures were always there in the images, waiting for someone to recognize their significance.
Paper Notes
Limitations
Imaging Challenges: The fluorescent labeling technique only lit up scattered individual neurons, not entire networks at once. This made it difficult to see both ends of a nanotube connection simultaneously and count exactly how many nanotubes exist throughout the brain. Think of it like trying to map a subway system when you can only see a few random stations at a time.
The team also reanalyzed existing microscopy images that were originally taken for other research purposes, so those images weren’t optimized for studying these specific structures.
Laboratory vs. Real Brain Conditions: Some experiments used neurons grown in laboratory dishes at reduced densities or studied very young developing neurons. Brain cells in a dish don’t behave exactly like cells in an intact, mature brain, so the findings may not fully reflect what happens in living animals or humans.
When injecting amyloid-beta proteins into single neurons, the researchers had to use pressure to get the proteins inside the cell. While control experiments suggested the nanotubes were doing the real transport work, the injection process itself could have forced some unnatural movement.
Protein Identification Issues: The antibody used to detect amyloid-beta in tissue samples also recognizes amyloid precursor protein (APP), the larger molecule from which amyloid-beta is cut. This means in some experiments, the researchers couldn’t tell whether they were seeing transported amyloid-beta or just the normal APP that was already inside cells.
Computer Model Simplifications: The computational simulations made several simplifying assumptions. They treated nanotube connections as fixed rather than constantly forming and disappearing. They also simplified how amyloid proteins clump together and how cells break down toxic proteins. The simulated disease timeline might not match exactly how Alzheimer’s progresses in living brains.
Scope of Study: This research focused mainly on one type of neuron (pyramidal cells) in the brain’s outer layer. Scientists don’t yet know whether other types of brain cells or other brain regions have similar nanotube networks. The molecular machinery that controls when nanotubes form and how they actually shuttle cargo between cells remains only partially understood.
What This Means: These limitations don’t invalidate the findings, but they do mean more research is needed to understand exactly how widespread these nanotubes are, how they work in intact brains, and whether they play the same role across different types of neurons and brain regions.
Funding and Disclosures
This work was supported by National Institutes of Health Grants DP1MH119428 and R01NS138176 to Hyung-Bae Kwon. The authors declared no competing interests. APP/PS1 mice were provided by Richard Huganir’s laboratory at Johns Hopkins University School of Medicine. All animal procedures were approved by the Johns Hopkins University Animal Care and Use Committee (protocol number MO22M170) and followed National Institutes of Health guidelines.
Publication Details
Chang M, Krüssel S, Parajuli LK, Kim J, Lee D, Merodio A, Kwon J, Okabe S, Kwon HB. “Intercellular communication in the brain through a dendritic nanotubular network.” Science, Volume 390, Issue 6717, eadr7403 (2025). DOI: 10.1126/science.adr7403. Published October 2, 2025.







