
Fatty foods start affecting your gut after just 48 hours. (Prostock-studio/Shutterstock)
In a nutshell
- Just two days of eating a high-fat diet can impair critical immune cells in the gut, weakening the intestinal barrier and increasing gut permeability.
- Saturated fats like those in butter and fatty meats suppress gut-protective immune responses, while unsaturated fats such as olive oil help preserve gut health.
- The damage from short-term high-fat eating is reversible. Returning to a regular diet can restore gut immune function and microbial balance within a week.
PARKVILLE, Australia — Your intestines register the impact of fatty foods within just 48 hours. New research from Australia reveals that high-fat meals rapidly disable critical immune cells that protect your intestinal barrier—not over months or years as previously thought, but in a matter of days. This discovery suggests our daily food choices have more immediate consequences than previously understood.
The Rapid Gut Response to High-Fat Diet
The study, published in the journal Immunity, reveals that consuming a high-fat diet quickly suppresses specialized immune cells called ILC3s (group 3 innate lymphoid cells), which produce a crucial protective substance called interleukin-22 (IL-22).
This rapid suppression of IL-22 creates a domino effect throughout the digestive system. IL-22 normally maintains your intestinal barrier by stimulating the production of antimicrobial peptides, mucus, and tight-junction proteins. These are all critical components that prevent harmful bacteria and toxins from leaking into your bloodstream. When high-fat diets suppress IL-22 production, the gut becomes more permeable, a condition often referred to as “leaky gut.”
“The more saturated fats we eat, the more inflammation that builds up,” says study author Cyril Seillet from The Walter and Eliza Hall Institute of Medical Research, in a statement. “But this inflammation build-up is initially silent, remaining hidden in our bodies until years later, where it can present as chronic inflammation.”
Not All Fats Have the Same Effects
The research team also discovered that different types of dietary fats have dramatically opposite effects on gut health. Saturated fatty acids—found in foods like butter, fatty meats, and palm oil—impaired immune cell function and increased intestinal inflammation. In contrast, unsaturated fatty acids—like those in olive oil and avocados—helped maintain normal IL-22 production and gut barrier function.
To investigate these effects, the scientists fed mice different diets for varying periods. Some received regular chow, while others consumed food with 36% or 60% fat content. The researchers observed that after just two days on high-fat diets, certain subtypes of ILC3 immune cells already showed reduced IL-22 production. By day seven, all ILC3 subtypes were compromised.
This dietary change simultaneously triggered rapid shifts in the gut microbiome, the community of bacteria living in our intestines. Within a week, beneficial bacteria that produce short-chain fatty acids declined, while potentially harmful species increased. Some of these harmful bacteria, like Enterococcus gallinarum, are known to reduce intestinal mucus and damage the connections between gut cells.
Using specialized equipment to measure different factors in the intestines, the researchers confirmed that after just one week on a high-fat diet, the mice’s intestines became measurably “leakier,” allowing more substances to pass through that would normally be kept out.
Reversing the Damage From Fatty Foods
These negative effects weren’t permanent. When mice were returned to a regular diet after a week of high-fat food, their gut function began to recover within just two days. By seven days back on normal food, most immune functions had normalized, suggesting dietary interventions could work quickly to restore gut health.
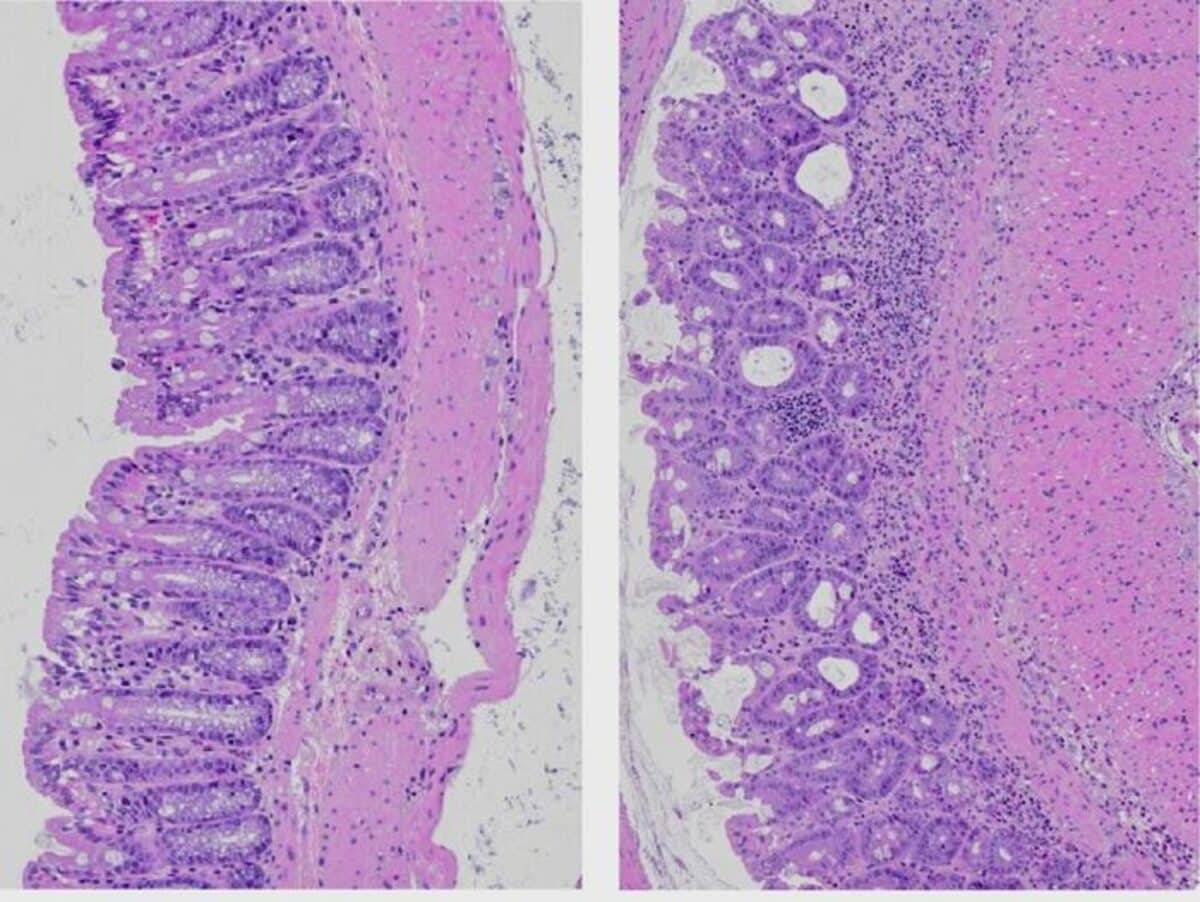
The researchers found that saturated and unsaturated fatty acids follow entirely different metabolic pathways in immune cells. Saturated fats like palmitic acid were processed through a pathway called fatty acid oxidation, which impaired immune function. Unsaturated fats like oleic acid formed protective lipid droplets in the cells, helping maintain proper immune responses.
To test how these dietary differences might affect recovery from intestinal inflammation, the researchers induced colitis in mice fed different diets. Those consuming saturated fats lost more weight and showed greater tissue damage than mice eating unsaturated fats or regular diets.
These findings could apply to humans as well. When the researchers isolated human immune cells and exposed them to different fatty acids in laboratory tests, they observed similar effects: saturated fats suppressed IL-22 production while unsaturated fats maintained normal immune function.
This could be why many people experience digestive discomfort shortly after periods of unhealthy eating, such as during holidays or vacations. Switching to a diet high in saturated fats can cause immediate changes in gut health, possibly setting the stage for long-term inflammatory diseases down the road.
This also may explain why Mediterranean diets rich in olive oil and other sources of unsaturated fats are associated with better gut health and lower inflammation. These dietary patterns aren’t just beneficial over a lifetime; they may provide immediate protection for intestinal function.
The gut doesn’t lie, and it doesn’t wait. What you eat today affects your intestinal health almost immediately, with different fats triggering entirely different immune responses. While your body can recover from dietary damage, the speed at which high-fat foods compromise gut protection suggests we should take our daily food choices more seriously than previously thought.
Paper Summary
Methodology
Researchers conducted a series of experiments using mice fed different diets, including regular diet (RD), high-fat diets (HFD) with 36% or 60% fat content, and specialized diets enriched with either palmitic acid (saturated) or oleic acid (unsaturated). The experiments primarily examined how these diets affected intestinal immune cells called ILC3s, which produce IL-22, a cytokine critical for gut barrier function. The researchers used flow cytometry to analyze immune cell populations, measured intestinal permeability using specialized Ussing chambers, performed genetic sequencing of gut bacteria, and conducted single-cell RNA sequencing to understand gene expression changes. They also isolated ILC3s and cultured them with different fatty acids to examine direct effects. Additionally, they induced colitis using dextran sodium sulfate (DSS) to test how different diets affected recovery from intestinal inflammation.
Results
Within 48 hours of high-fat diet exposure, mice showed impaired IL-22 production by ILC3s, which became more pronounced by day 7. This coincided with increased gut permeability, decreased expression of antimicrobial peptides, mucus, and tight junction proteins. The gut microbiome rapidly shifted, with beneficial bacteria declining and potentially harmful species increasing. When mice returned to a regular diet, these changes were largely reversed within 7 days. Importantly, saturated and unsaturated fatty acids had opposing effects. Saturated fats like palmitic acid reduced IL-22 production through fat oxidation pathways, while unsaturated fats like oleic acid maintained IL-22 production by forming protective lipid droplets in immune cells. When inflammation was induced, mice fed saturated fat diets showed worse outcomes than those on unsaturated fat or regular diets. Similar effects were observed in human immune cells exposed to different fatty acids.
Limitations
The study primarily used mouse models, so the findings may not fully translate to humans despite similar effects observed in cultured human cells. The research focused on short-term high-fat diet exposure rather than long-term effects. The study didn’t explore how these dietary changes might interact with pre-existing health conditions or genetic factors that influence individual responses to high-fat diets. The authors note that the precise molecular mechanisms connecting fatty acid metabolism to IL-22 inhibition require further investigation. Additionally, the study doesn’t address whether dietary supplements or interventions might prevent the negative effects of high-fat diets.
Funding/Disclosures
This work was supported by grants and fellowships from the National Health and Medical Research Council (NHMRC) of Australia (grants 1165443, 1123000, and 2008090 to C.S.; 1155342 and 2026084 to S.L.N.; and 2025614 to S.Z.) and Melbourne Research Scholarship (to L.X.). The Walter and Eliza Hall Institute is supported through Victorian State Government Operational Infrastructure Support and Australian Government NHMRC IRIIS. The authors declared no competing interests.
Publication Information
The paper titled “Acute exposure to high-fat diet impairs ILC3 functions and gut homeostasis” was published in the journal Immunity (Volume 58, pages 1185-1200) on May 13, 2025. The first author is Le Xiong, and the corresponding author is Cyril Seillet from Monash University in Australia. The study was conducted by researchers from multiple Australian institutions, including The Walter and Eliza Hall Institute of Medical Research, University of Melbourne, Monash University, Baker Heart and Diabetes Institute, and La Trobe University.







