
Beach erosion after storm activity along Australia's Gold Coast (© DavidJMorgan - stock.adobe.com)
CHICAGO — As coastal communities around the world grapple with the growing threat of erosion, researchers have uncovered a surprising potential solution: electricity. Scientists from Northwestern University say that applying mild electrical currents to marine sands could create natural, sustainable defenses against the relentless assault of waves and rising sea levels.
Coastal erosion is a pressing global issue, with approximately 40% of the world’s population living in coastal areas. As sea levels rise and storms intensify, nearly 26% of the Earth’s beaches could vanish by the end of this century. Traditional methods to combat this problem, such as seawalls and beach replenishment, often provide only temporary relief and can be costly to maintain.
Now, Northwestern’s Alessandro Rotta Loria and his team have found a way to turn sand into a rock-like substance, creating a natural armor for our coasts that could last for generations.
“My aim was to develop an approach capable of changing the status quo in coastal protection — one that didn’t require the construction of protection structures and could cement marine substrates without using actual cement,” Rotta Loria says in a statement. “By applying a mild electric stimulation to marine soils, we systematically and mechanistically proved that it is possible to cement them by turning naturally dissolved minerals in seawater into solid mineral binders — a natural cement.”
This innovative technique, called electrodeposition, draws inspiration from nature’s own architects: clams, mussels, and other shell-dwelling sea creatures. Just as these marine mollusks use dissolved minerals in seawater to construct their protective homes, the researchers harnessed these same minerals to create a natural cement between grains of sand. The twist? Instead of relying on the slow process of metabolic energy, they used electricity to kickstart the reaction.

Samples of seawater-soaked sand in Rotta Loria’s laboratory. The vertical, silver posts are electrodes. (Credit: Northwestern University)
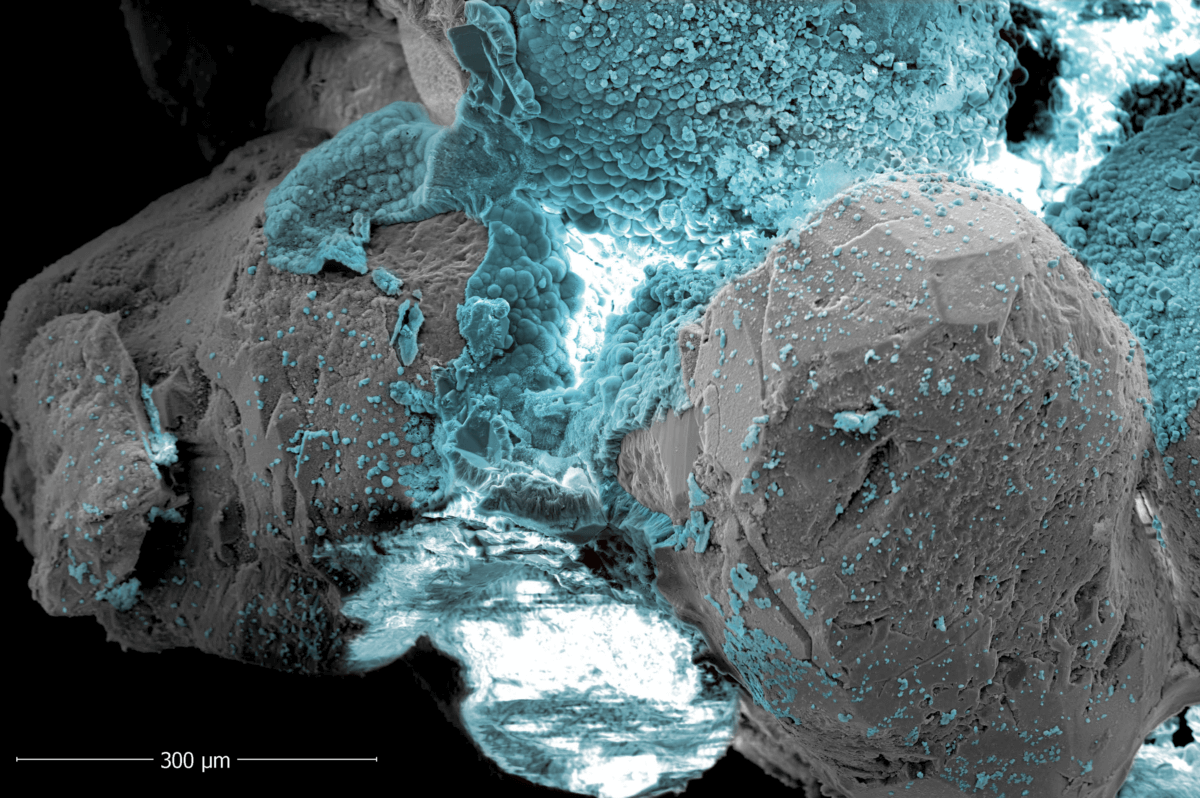
In their laboratory experiments, the team applied mild electrical currents (ranging from 2 to 4 volts) to seawater-saturated sand. The results, published in Communications Earth & Environment, were nothing short of miraculous. The electricity triggered chemical reactions that converted naturally occurring ions and minerals in the seawater into solid calcium carbonate—the same stuff seashells are made of—and other mineral compounds like magnesium hydroxide and hydromagnesite.
These newly formed minerals act like a super-glue, binding sand particles together into a solid, immovable mass. “After being treated, the sand looks like a rock,” Rotta Loria explains. “It is still and solid, instead of granular and incohesive. The minerals themselves are much stronger than concrete, so the resulting sand could become as strong and solid as a sea wall.”
The researchers conducted a series of laboratory experiments using custom-designed electrochemical cells filled with silica sand and artificial seawater. By varying the voltage applied, the density of the sand, and the duration of the electrical treatment, they were able to observe how different conditions affected the formation and distribution of mineral deposits within the sand.

The results were striking. Depending on the voltage applied, the researchers observed the formation of different types of minerals. At lower voltages, calcium carbonate (the same material found in seashells and coral reefs) was the primary deposit. As the voltage increased, magnesium hydroxide became more prevalent. Surprisingly, the team also discovered the formation of hydromagnesite, a mineral not previously observed in similar electrodeposition studies.
These mineral deposits didn’t just coat the sand particles; they formed bridges between them, effectively creating a cohesive, rock-like material from what was once loose sand. The strength of these newly formed “rocks” varied based on the amount of mineral content, with some samples achieving strengths of several megapascals – comparable to weak concrete.
Perhaps most importantly, the electrodeposition process significantly reduced the sand’s permeability. In some cases, the hydraulic conductivity (a measure of how easily water can flow through the material) decreased by an order of magnitude. This could have profound implications for mitigating erosion, as it would make the treated sand much more resistant to water infiltration and the undermining effects of waves and currents.
The implications of this discovery are vast. Traditional methods of coastal protection, such as building sea walls or injecting cement into the ground, are not only expensive but often temporary solutions. Sea walls themselves can erode, and the sand beneath them can liquefy, causing these multi-million dollar structures to sink into the very ground they’re meant to protect. In contrast, the electrodeposition method works with nature, strengthening the existing beach structure from within.
What’s more, this process is remarkably cost-effective. The researchers estimate that electrically cementing a cubic meter of sand would cost between $3 to $6, compared to up to $70 for conventional methods using binders. It’s also environmentally friendly and reversible—if needed, the process can be undone by simply switching the electrodes.
But perhaps the most exciting aspect of this research is its potential for widespread application. Beyond strengthening beaches, this technique could be used to stabilize sand dunes, reinforce seabeds beneath existing structures, and even heal cracks in reinforced concrete infrastructure along the shore.

However, as with any new technology, there are challenges to overcome before this method can be widely implemented. The researchers note that the process is sensitive to variables such as sand density and ionic concentration in the seawater. Scaling up from laboratory conditions to real-world beaches will require further study and refinement.
Despite these hurdles, the promise of this approach is clear. As climate change continues to threaten coastal areas with rising sea levels and more frequent storms, innovative solutions like electrodeposition could play a crucial role in protecting our shores. By harnessing the power of electricity and the minerals naturally present in seawater, we may have found a way to work with nature, rather than against it, in our ongoing battle against coastal erosion.
Paper Summary
Methodology
The researchers conducted their experiments using custom-designed glass cells filled with various types of sand (including silica, calcareous, and iron sands) and artificial seawater. They applied different voltages (2.0, 3.0, and 4.0 volts) to the sand using electrodes, varying the duration of the electrical treatment. Throughout the experiments, they monitored pH levels and electrical current. After treatment, the researchers used various analytical techniques, including scanning electron microscopy, X-ray diffraction, and thermogravimetric analysis, to examine the type, amount, and distribution of minerals formed in the sand. They also conducted tests to measure the strength and permeability of the treated sand.
Results
The study found that applying electrical current to seawater-saturated sand resulted in the formation of mineral deposits that cemented sand particles together. Lower voltages (2.0-3.0V) primarily produced calcium carbonate, while higher voltages (4.0V) favored magnesium hydroxide and hydromagnesite formation. The treated sand showed significantly increased strength, with some samples becoming as solid as weak concrete. The process also greatly reduced the sand’s permeability, making it more resistant to water infiltration. The volume of sand affected by the treatment increased with voltage and duration, with longer treatments affecting larger areas.
Limitations
While promising, this study has several limitations. The experiments were conducted under controlled laboratory conditions, which may not fully replicate the complex environment of real beaches. The longest duration tested was 28 days, which may not be sufficient to understand long-term effects and stability of the mineral formations. The study used artificial seawater, which might not represent all coastal environments. Additionally, while the researchers predict the treatment should last for decades, long-term durability in real-world conditions has not yet been tested.
Discussion and Takeaways
This research presents a novel approach to coastal erosion mitigation that could be more sustainable, cost-effective, and adaptable than traditional methods. The ability to selectively produce different minerals by controlling voltage offers flexibility in tailoring the treatment to specific coastal environments. The reduction in sand permeability and increase in strength suggest that this method could significantly enhance beach resistance to erosion. The reversibility of the process and its potential eco-friendliness are additional advantages. However, the researchers emphasize that more work is needed to understand how this process would work in real-world conditions and at larger scales.
Funding and Disclosures
The study was funded by several organizations, including the Army Research Office and the Center for Engineering Sustainability and Resilience at Northwestern University. The researchers used facilities at Northwestern University’s NUANCE Center, IMSERC Crystallography facility, and the Jerome B. Cohen X-Ray Diffraction Facility, which receive support from various sources including the National Science Foundation. The authors declared no competing interests related to the study.







