
(Photo by sasirin pamai on Shutterstock)
In A Nutshell
- The small, short trial showed no serious side effects, but much larger and longer studies are needed to prove the drug is safe and actually prevents heart disease
- An experimental drug called TLC-2716 reduced triglycerides by roughly 40% overall and over 60% in people with higher baseline levels after just 14 days of treatment
- The drug targets fat production in the liver and gut while avoiding problems that derailed earlier attempts to develop similar medications
- Remnant cholesterol, a dangerous blood fat linked to heart attacks, dropped by about 61% after meals in people taking higher doses
Millions of Americans take statins to lower their cholesterol, but many still have dangerously high levels of triglycerides and other blood fats that statins don’t address. Now, an experimental pill has reduced these hard-to-treat fats by up to 60% in a small early-stage trial, offering potential hope for people whose heart disease risk remains high despite standard treatments.
The drug, called TLC-2716, targets a molecular switch in the liver that controls fat production. After just 14 days of daily pills, participants saw notable drops in triglycerides and remnant cholesterol, a particularly nasty type of blood fat that has been linked to clogged arteries and heart attacks, even when LDL cholesterol is under control.
What stood out was how well the drug worked for people who started with higher triglycerides. Among volunteers with levels above 100 mg/dL (already creeping toward problem territory), the highest dose dropped these fats by more than 60%. That’s a substantial change for such a short study period.
The study, published in Nature Medicine, represents a step forward for a class of drugs that researchers have been trying to perfect for decades. Earlier versions caused worrisome side effects because they affected the entire body. This one stays mostly in the liver and gut, where it blocks fat production and absorption without interfering with the heart’s natural cholesterol-clearing mechanisms.
Why Previous Drugs Failed
The liver has molecular switches called liver X receptors that tell it whether to make more fat or less. When these switches flip on, fat production ramps up. When they flip off, production slows down. Scientists realized that blocking these switches could lower dangerous blood fats, but early drugs that did this throughout the whole body ran into problems.
The main concern was that blocking these switches everywhere might stop the body from clearing cholesterol out of arteries, potentially making heart disease worse instead of better. White blood cells normally use these same switches to remove excess cholesterol from artery walls and transport it back to the liver for disposal.
TLC-2716 solves this issue by staying put. After the pill is swallowed, the liver quickly absorbs it. Very little enters the general bloodstream. Researchers confirmed this by measuring genes in white blood cells and found the drug had virtually no effect on them, even while it was busy shutting down fat production in the liver and reducing fat absorption in the intestines.
Three Ways to Attack the Problem
The drug works through three routes at once. First, it tells the liver to stop making so many fat molecules in the first place. Second, it reduces how much dietary fat the intestines absorb from food. Third, it removes the brakes from enzymes that normally break down fat particles floating in the bloodstream.
The body has natural fat-clearing enzymes, but it also has proteins that hold them back (otherwise they’d work too well). TLC-2716 lowers levels of these brake proteins, essentially letting the body’s natural “cleanup crew” work more efficiently.
The results went beyond just triglycerides. People taking the two highest doses also saw big drops in remnant cholesterol. You’ve probably heard about “good” HDL and “bad” LDL cholesterol, but remnant cholesterol is the leftover junk that forms after your body processes triglyceride-rich particles. It’s been linked to clogged arteries and heart attacks, and many people have high levels of it even when their LDL looks fine on a standard cholesterol test.
Testing Safety Before Human Trials
Before giving TLC-2716 to people, researchers spent years testing it in animals and lab-grown human liver tissue. The drug consistently lowered fat levels and reduced inflammation. They tested it for up to 26 weeks in mice and 4 weeks in monkeys at doses higher than what humans would take, and the short-term studies did not raise major safety concerns.
In the human trial, which lasted only 14 days, the drug appeared well tolerated. The most common side effects were things like headache, diarrhea, and mild stomach discomfort, and these happened at similar rates in people taking the placebo. No serious side effects were reported in this small, short study, though longer trials will be needed to fully assess safety.
A Daily Pill, Not a Monthly Shot
Several newer drugs for high triglycerides require monthly injections and can be expensive. TLC-2716 is just a pill you’d take once a day, which could make it easier to use and potentially more accessible if it reaches the market.
The researchers reported that the drug clears from the bloodstream relatively quickly after dosing, which may be safer in the long run since it doesn’t appear to accumulate in the body over time.
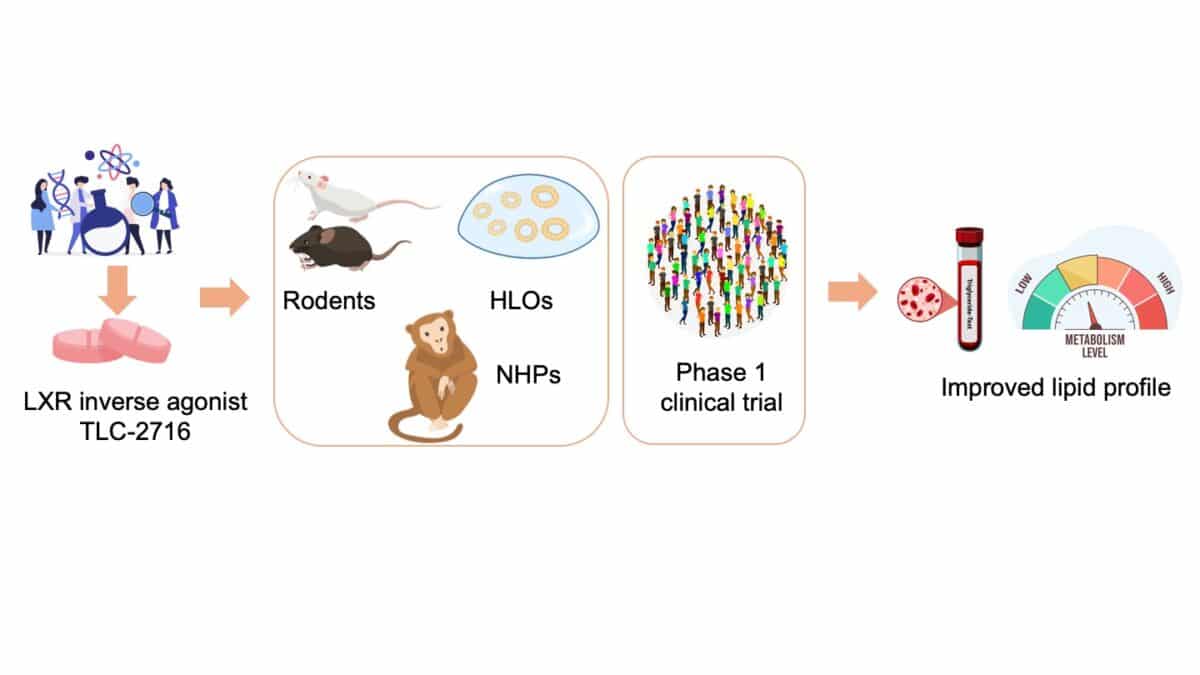
What Happens Next
This was a phase 1 trial, designed mainly to test safety, not cure disease. The 100 volunteers were healthy people with mostly normal cholesterol, not patients struggling with severe triglyceride problems. Even so, the drug showed clear benefits in just two weeks.
Now researchers are testing it in people who actually need it: those with obesity, fatty liver disease, and triglycerides above 350 mg/dL. These patients have few good options beyond diet changes and older drugs that don’t work very well. If TLC-2716 proves safe and effective in this group, it could fill a major gap.
The bigger question is whether lowering these fats actually prevents heart attacks and strokes. That’s the test every cholesterol drug eventually faces, and it takes years to answer. Statins clearly help save lives. Newer triglyceride drugs are still proving themselves. TLC-2716 will need to do the same.
For now, though, these findings represent real progress. Researchers have been trying to develop drugs like this for decades, and most attempts failed because they caused problems in other parts of the body. This version stayed where it should, worked as intended, and didn’t cause serious side effects. That’s a major step forward for scientists, and reason for optimism among people whose abnormally high blood fats put them at risk despite taking statins.
Disclaimer: This article discusses early-stage research on an experimental drug that has not been approved for medical use. The findings are from a small, short-term phase 1 trial in healthy volunteers and do not prove the drug is safe or effective for treating disease. Readers should not change their medications or treatment plans based on this information. Always consult with a healthcare provider before making any decisions about medical treatment.
Paper Summary
Limitations
The phase 1 trial was conducted in healthy volunteers with relatively normal baseline lipid levels, limiting the ability to fully assess the drug’s potential in people with severe dyslipidemia. The study duration was only 14 days, providing no information about long-term efficacy or safety. The small sample sizes in each dose group (8 active and 2 placebo per cohort) limited statistical power for detecting modest effects or uncommon side effects. The study did not assess cardiovascular outcomes or determine whether lipid reductions translate to reduced heart attack and stroke risk. The trial was conducted at a single site in New Zealand with limited ethnic diversity, and results may not generalize to other populations.
Funding and Disclosures
This research was supported by OrsoBio (Menlo Park, CA), the employer of several study authors. Additional support came from the École Polytechnique Fédérale de Lausanne, National Institutes of Health grants, the Japan Agency for Medical Research and Development, and other Japanese funding sources. Multiple authors are employed by or serve as advisors to OrsoBio, and one author is employed by WM Therapeutics. The pharmaceutical company relationship raises potential for bias in study design, interpretation, and reporting of results, though the independent academic collaborators and publication in a peer-reviewed journal provide some mitigation of these concerns.
Publication Details
Xiaoxu Li, Giorgia Benegiamo, Archana Vijayakumar, Natalie Sroda, Masaki Kimura, Ryan S. Huss, Steve Weng, Eisuke Murakami, Brian J. Kirby, Giacomo V. G. von Alvensleben, Claus Kremoser, Edward J. Gane, Takanori Takebe, Robert P. Myers, G. Mani Subramanian, and Johan Auwerx. “An oral, liver-restricted LXR inverse agonist for dyslipidemia: preclinical development and phase 1 trial.” Published January 16, 2026 in Nature Medicine. DOI: 10.1038/s41591-025-04169-6. The study was conducted from July 27, 2022, to June 18, 2023, at Auckland Clinical Research in New Zealand and registered at ClinicalTrials.gov (NCT05483998). Authors represent institutions including École Polytechnique Fédérale de Lausanne (Switzerland), OrsoBio (United States), Cincinnati Children’s Hospital Medical Center (United States), WM Therapeutics (Germany), University of Auckland (New Zealand), and Osaka University (Japan).







