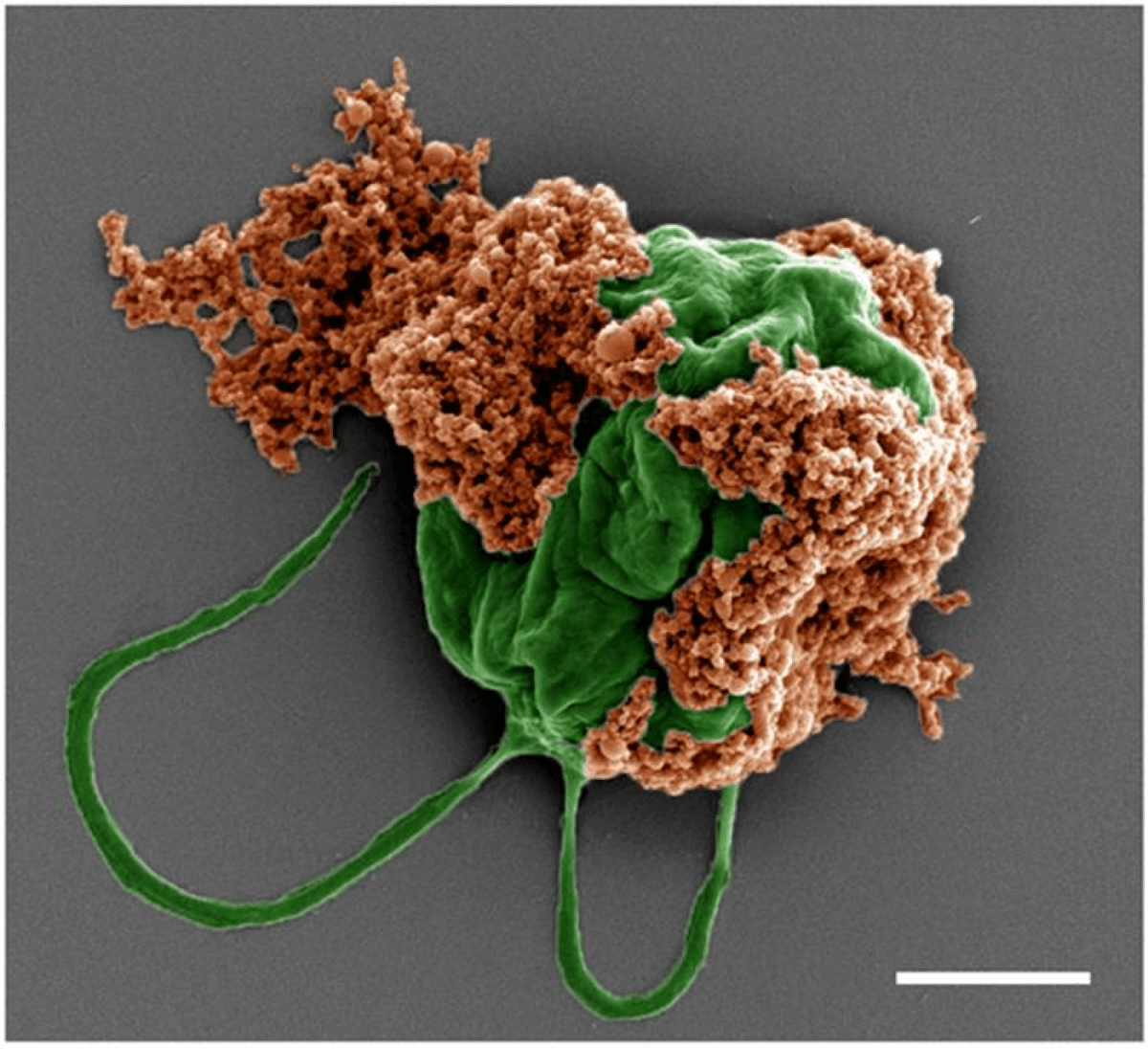
Colored SEM image of a microrobot made of an algae cell (green) covered with drug-filled nanoparticles (orange) coated with red blood cell membranes. Scale bar: 2 µm. (CREDIT: Zhengxing Li)
SAN DIEGO — In the relentless battle against cancer, researchers are constantly searching for new and innovative ways to deliver life-saving treatments while minimizing the collateral damage to healthy tissues. One of the most challenging aspects of cancer therapy is the delivery of drugs to metastatic tumors, which can spread far from the primary site and take root in difficult-to-reach areas of the body. This is particularly true for lung cancer, where the complex anatomy and delicate structure of the lungs can make targeted drug delivery a daunting task.
Enter microalgae robots, an innovative new technology that combines the power of nature with the precision of nanotechnology to create a drug delivery system like no other. These tiny, bio-hybrid machines consist of living microalgae, single-celled organisms known for their ability to swim through biological fluids, paired with cancer-fighting nanoparticles that are loaded with chemotherapy drugs and cloaked in a protective layer of red blood cell membranes.
According to the details published in Science Advances, the beauty of this system lies in its elegance and simplicity. The microalgae, a species called Chlamydomonas reinhardtii, serve as the “engine” of the robot, propelling it through the fluid-filled spaces of the lungs with their natural swimming motion. The nanoparticles, made of a biodegradable polymer and coated with a layer of red blood cell membranes, act as the “cargo hold,” carrying the chemotherapy drug doxorubicin (DOX) safely through the body’s immune defenses and releasing it directly at the site of the metastatic tumors.
However, the real magic of the microalgae robots lies in their ability to navigate the complex terrain of the lungs and penetrate deep into the tissue, delivering their cancer-fighting payload to even the most hard-to-reach tumors. This is thanks to the microalgae’s innate ability to sense and respond to their environment, as well as their uncanny knack for evading the lungs’ natural defense mechanisms, such as alveolar macrophages that would normally engulf and destroy foreign particles.
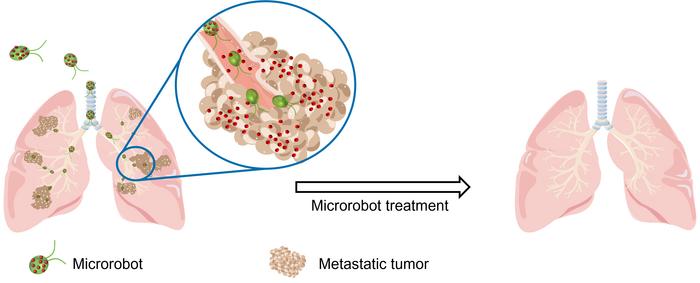
For cancer patients, particularly those battling the deadly scourge of lung cancer, the implications of this technology are nothing short of revolutionary. By delivering chemotherapy drugs directly to the site of the metastatic tumors, the microalgae robots have the potential to significantly enhance the effectiveness of treatment while simultaneously reducing the risk of side effects and toxicity to healthy tissues. This could lead to better outcomes, longer survival times, and an improved quality of life for patients undergoing what is often a grueling and debilitating course of treatment.
Moreover, the fact that the microalgae robots can be administered directly into the lungs via a simple intratracheal injection means that patients could potentially receive their treatments on an outpatient basis without the need for lengthy hospital stays or invasive surgical procedures. This not only makes the treatment more convenient and accessible but also reduces the burden on patients and their families during what is already an incredibly challenging time.
Of course, as with any new technology, there is still much work to be done before microalgae robots can become a standard part of the cancer treatment arsenal. Further research is needed to fully understand the mechanisms behind their enhanced performance, as well as to validate their safety and efficacy in human clinical trials. But the early results are incredibly promising, and the potential impact on the lives of cancer patients cannot be overstated.
With cancer continuing to reign as one of the leading causes of death and suffering, the development of new therapies like microalgae robots offers a glimmer of hope on the horizon. By harnessing the power of nature and combining it with the cutting edge of scientific research, we may be on the cusp of a new era in cancer treatment, one in which even the most devastating forms of the disease can be effectively targeted and eliminated. For the millions of patients and families touched by cancer each year, that hope cannot come soon enough.
“The active swimming motion of the microrobots significantly improved distribution of the drug to the deep lung tissue, while prolonging retention time,” says study co-first author Zhengxing Li in a media release. “This enhanced distribution and prolonged retention time allowed us to reduce the required drug dosage, potentially reducing side effects while maintaining high survival efficacy.”
Paper Summary
Methodology
To create these tiny cancer-fighting machines, the researchers first loaded the chemotherapy drug doxorubicin (DOX) into biodegradable polymer nanoparticles. They then coated these drug-laden nanoparticles with red blood cell membranes, which act as a cloak to shield the cargo from the body’s immune system. The final step involved attaching these nanoparticles to the surface of green microalgae, Chlamydomonas reinhardtii, using a chemical linking process called click chemistry.
The choice of microalgae as the “engine” for these microrobots was no accident. These tiny, single-celled organisms are not only excellent swimmers, capable of propelling themselves through biological fluids, but they also possess a natural fluorescence that allows researchers to track their movements. Additionally, microalgae are biocompatible and have been deemed safe for use by the FDA.
To test the effectiveness of their microalgae-robot drug delivery system, the researchers administered the bots directly into the lungs of mice via intratracheal injection. They compared the performance of their microrobots against free DOX and DOX-loaded nanoparticles without the microalgae component.
Results
The results were nothing short of remarkable. The microalgae robots exhibited significantly enhanced retention and distribution within the lungs compared to the free drug and nanoparticle controls. At 24 hours post-administration, the lungs of mice treated with the microrobots contained 4.4 times more DOX than those treated with the nanoparticles alone.
Microscopic imaging of lung tissue sections revealed that the microrobots were able to penetrate deep into the lungs, delivering their drug payload to hard-to-reach areas. In contrast, the nanoparticles without the microalgae component showed limited penetration and distribution.
The researchers attributed this enhanced performance to the microalgae’s ability to actively swim and evade uptake by alveolar macrophages, the lungs’ resident immune cells responsible for clearing foreign particles. Flow cytometry analysis confirmed that the microalgae robots were indeed able to avoid macrophage engulfment for extended periods, allowing them to continue delivering their drug cargo.
In a mouse model of melanoma lung metastasis, treatment with the microalgae robots resulted in a dramatic reduction in tumor burden and a significant increase in survival compared to the control groups. Mice treated with the microrobots had a median survival time of 37 days, a 40% increase over the 27-day median survival of mice receiving free DOX or DOX-loaded nanoparticles.
Limitations
While the results are undeniably promising, the researchers acknowledge that there are still some limitations to their study. For one, the exact mechanisms by which the microalgae robots enhance drug retention and antitumor efficacy need to be further elucidated. Factors such as bioadhesion, size effects, and clearance rates will need to be investigated in more detail to fully understand how the robots achieve their impressive lung penetration.
Additionally, the study was conducted in mouse models, and while these are valuable tools for preclinical research, the results will need to be validated in human clinical trials before the microalgae robots can be considered a viable treatment option for lung cancer patients.
Takeaways
This study represents a significant advancement in the field of targeted cancer therapy. By leveraging the unique properties of living microalgae, the researchers have created a drug delivery system that can overcome many of the limitations of traditional chemotherapy.
The ability of the microalgae robots to actively swim and evade the body’s natural clearance mechanisms is a game-changer. It means that more of the drug can reach its intended target, potentially leading to better treatment outcomes and fewer side effects.
Moreover, the fact that the microrobots can be administered directly into the lungs via intratracheal injection is a major advantage. This localized delivery approach minimizes systemic exposure to the drug, further reducing the risk of adverse effects on healthy tissues.
Looking ahead, the researchers envision that their microalgae robot platform could be further enhanced by incorporating additional targeting strategies, such as magnetic guidance or ultrasound trapping. These could allow for even more precise drug delivery to specific tumor sites.
While there is still much work to be done, the microalgae robots represent a promising new weapon in the ongoing battle against one of humanity’s most formidable foes.







