
Responsible for about 50,000 American deaths annually, pancreatic cancer is considered one of the most stubborn forms of the disease. (Credit: Jo Panuwat D on Shutterstock)
Akin to ‘taking a crowbar’ to the deadly disease, the drug pries cancer-growing proteins apart.
In A Nutshell
- Researchers created a drug called 164B8 that works like a molecular crowbar, physically prying apart and destroying a cancer-fueling protein called Pin1 instead of just blocking it.
- In mice with pancreatic cancer, the drug slashed tumor size by more than half and targeted both cancer cells and the dense support tissue that helps tumors resist treatment.
- The compound’s improved stability in the bloodstream and ability to penetrate tumors make it a promising candidate for future clinical trials against pancreatic and other gastrointestinal cancers.
- While early results are encouraging, the drug has only been tested in immunocompromised mice for two weeks, so longer studies in animals with normal immune systems are needed before human trials can begin.
A promising experimental drug works differently than standard cancer treatments. What makes it so unique? Instead of simply blocking a cancer-fueling protein, it physically pries the protein apart until it breaks down. Early tests in mice with pancreatic cancer showed the compound cut tumor growth by more than half.
Researchers at the University of California, Riverside developed a chemical called 164B8 that targets Pin1, a protein acting as a master switch for aggressive cancer growth. The drug binds to Pin1 and forces it into an unstable shape, causing the protein to fall apart. When tested in mice with spreading pancreatic cancer, the compound dramatically reduced tumor size compared to untreated animals.
Lead researcher Maurizio Pellecchia’s team designed the molecule to create structural instability in Pin1. The compound attaches permanently to a specific spot on the protein and exposes normally hidden regions. This triggers cells to recognize Pin1 as damaged and destroy it.
Pancreatic cancer kills roughly 50,000 Americans each year and is projected to become the second-leading cause of cancer deaths by 2030. Most patients survive less than a year after diagnosis because the disease resists chemotherapy, targeted drugs, and immunotherapy. Dense, fibrous tissue surrounding pancreatic tumors creates a barrier that shields cancer cells from most treatments.
How the Molecular Crowbar Works
Pin1 changes the shape of other proteins, activating cancer-promoting signals while shutting down tumor suppressors. Higher Pin1 levels correlate with rapid disease progression and poor survival in lung, oral, and pancreatic cancers.
Developing drugs against Pin1 has been challenging. Previous compounds either lacked potency, broke down too quickly in the bloodstream, or failed to actually degrade the protein inside cells.
Pellecchia’s team took a different approach. Rather than simply blocking Pin1, they engineered 164B8 to bind permanently to a specific spot on the protein and act as a molecular crowbar. The drug wedges into Pin1’s structure and forces two regions out of their normal positions. This exposes a water-repelling part of the protein that’s usually buried inside.
Cells detect this malformation and trigger their quality control systems to destroy the damaged protein—without requiring the complex tagging machinery that many other protein-destroying drugs depend on.
Stability Breakthrough Enables Testing
A key advance came from improving the drug’s stability. Earlier versions broke down rapidly in mouse blood. By adding a methyl group to the molecule, the team created 164B8, which lasted longer in the bloodstream and could better cross cell membranes.
Researchers tested 164B8 in laboratory dishes against three pancreatic cancer cell lines and against support cells isolated from patients with gastrointestinal tumors. Within 24 hours, the compound reduced Pin1 levels by 30-50% across all cell types. After removing the drug, Pin1 levels gradually climbed back to roughly half of normal within 48 hours.
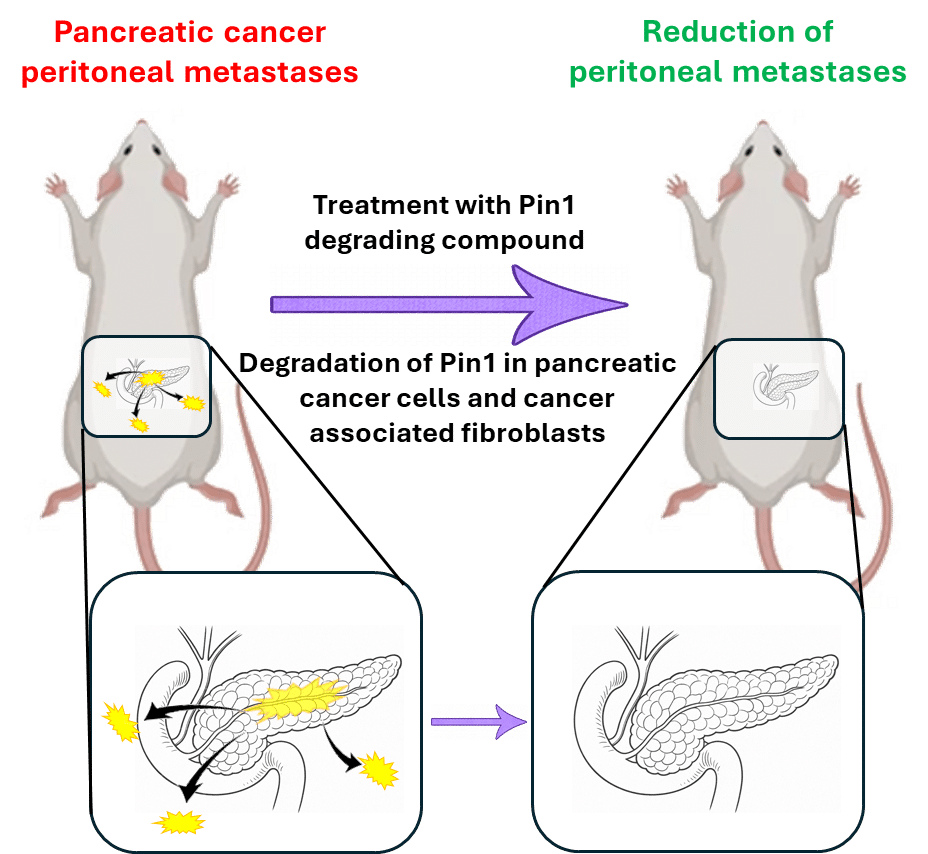
For the animal study, published in Molecular Therapy: Oncology, scientists injected 100,000 pancreatic cancer cells into the abdominal cavities of immunocompromised mice. Three days later, mice received either a placebo, 30 mg/kg of 164B8, or 60 mg/kg of 164B8 daily for two weeks.
Tumor growth was tracked using bioluminescence imaging since the cancer cells had been modified to glow. Both drug-treated groups showed drastically reduced tumor size compared to controls, with the higher dose producing the greatest effect. Blood tests revealed mild anemia and moderate platelet reduction in treated mice, possibly reflecting Pin1’s normal biological roles. The 30 mg/kg dose was well tolerated with minimal side effects.
Attacking Tumors and Their Support Structure
Pin1 appears overexpressed not only in cancer cells but also in the connective tissue cells that form dense scaffolding around pancreatic tumors. This barrier helps block immune cells and drugs from reaching cancer cells.
When researchers tested 164B8 against support cells isolated from three patients with spreading cancers from the biliary tract, appendix, and colon, the compound successfully degraded Pin1 across all cell types. Targeting Pin1 in both tumor cells and surrounding tissue could address multiple aspects of treatment resistance simultaneously.
To verify the drug reached tumors in living animals, researchers measured 164B8 concentrations six hours after the final dose. Tumors contained higher drug levels than adjacent normal tissue or liver in mice receiving the higher dose—average tumor concentrations reached 8 nanograms per milligram compared to 3.65 in nearby healthy tissue and 0.75 in liver.
Mice genetically engineered to lack Pin1 develop normally and remain healthy but show remarkable resistance to cancer formation. In humans, genetic variations that reduce Pin1 expression correlate with lower cancer incidence. These findings suggest temporarily depleting Pin1 with drugs might slow tumor growth without causing severe toxicity.
The crowbar mechanism works differently than PROTAC drugs, which have gained attention recently. PROTACs connect a target protein to cellular degradation machinery through a long linker molecule. 164B8 instead exploits the cell’s natural quality control pathways by making Pin1 look structurally defective—a simpler design that may offer manufacturing and medical advantages.
Limitations include use of immunocompromised mice and the relatively short two-week treatment window. Testing in other cancer types, larger animal models, and eventual human trials will eventually determine whether the approach benefits patients facing one of cancer medicine’s most stubborn diseases.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. If you have concerns about cancer or treatment options, please consult with qualified healthcare professionals.
Paper Notes
Limitations
The study used immunocompromised NSG mice that lack functional T cells, B cells, and natural killer cells, which limits assessment of potential immunological effects of Pin1 inhibition. The treatment duration of two weeks is relatively short and may not reflect long-term tolerability or the development of resistance mechanisms. The peritoneal metastasis model, while clinically relevant for pancreatic cancer, does not capture the full complexity of orthotopic pancreatic tumors or spontaneous metastasis.
Cell viability and efficacy studies were conducted primarily in mouse-derived KPC cells and two human pancreatic cancer cell lines, which may not represent the full heterogeneity of human pancreatic ductal adenocarcinoma. The cancer-associated fibroblast studies used cells from only three patients with different tumor types, limiting generalizability.
Complete blood counts showed mild anemia and moderate thrombocytopenia in treated mice, suggesting potential hematologic toxicity that would require monitoring in clinical development. Two mice in the 60 mg/kg group developed ascites and required early euthanasia, though carcinomatosis was not observed, leaving the cause uncertain. Pharmacokinetic parameters such as half-life, clearance, and area under the curve were not reported. The study did not compare 164B8 to current standard-of-care treatments for pancreatic cancer or evaluate combination therapy approaches.
Funding and Disclosures
Financial support was obtained from National Institutes of Health grants NS107479, CA168517, CA242620, and CA285114. Maurizio Pellecchia holds the Daniel Hays Chair in Cancer Research at the School of Medicine at the University of California, Riverside. Maurizio Pellecchia is a co-founder of Armida Labs, Inc. in Riverside, California. Maurizio Pellecchia, Parima Udompholkul, and Giulia Alboreggia are listed as possible co-inventors on a patent application filed by the University of California, Riverside, related to the compounds described in the application. No other competing interests were declared.
Publication Details
Alboreggia G, Li T, Prentiss AM, Udompholkul P, Xia F, Synold T, Wu J, Feng M, Raoof M, Pellecchia M. “Pre-clinical evaluation of a potent and effective Pin1-degrading agent in pancreatic cancer,” Molecular Therapy: Oncology. 2025 Dec;33. DOI:10.1016/j.omton.2025.201078. Published online November 13, 2025.







