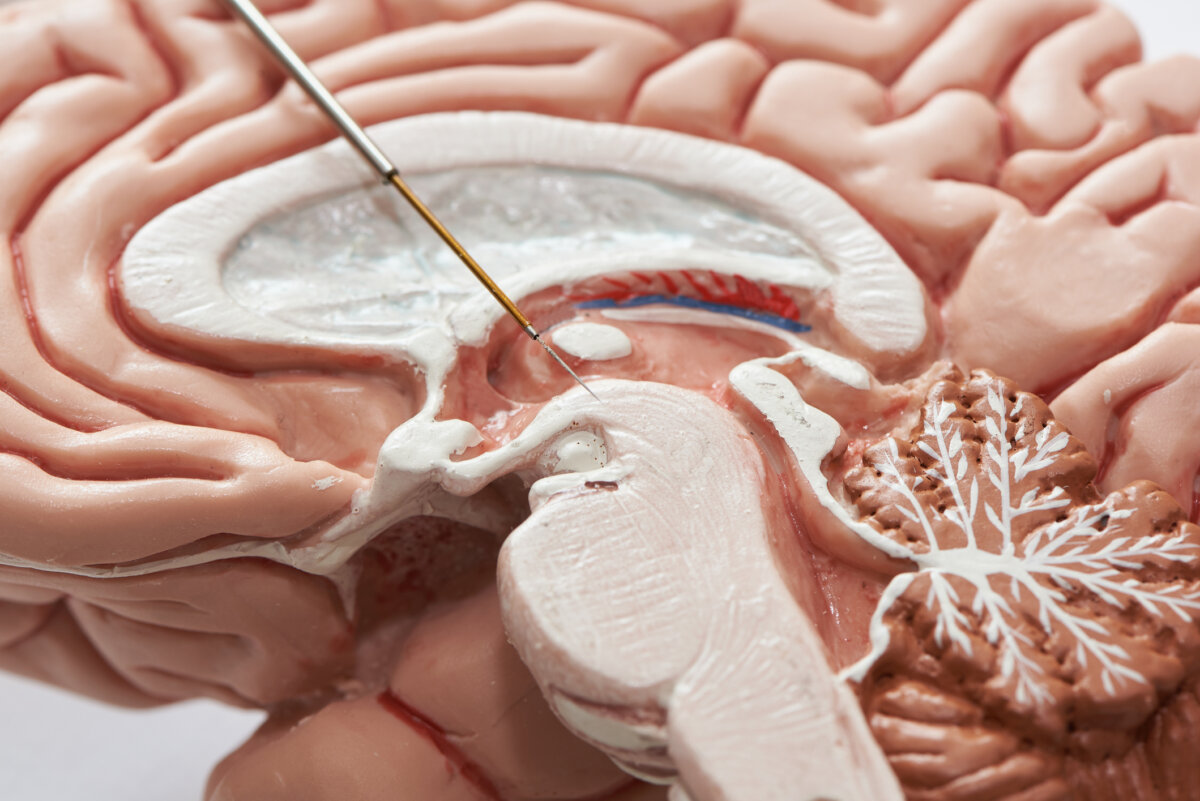
Close-up view of microelectrode recording on the brain model, in subthalamic nucleus for Parkinson's disease surgery. (Credit: © Teeradej Srikijvilaikul | Dreamstime.com)
‘Wireless’ deep brain stimulation technique offers promising potential for neurodegenerative diseases
BEIJING — Parkinson’s disease, which affects millions worldwide and is second only to Alzheimer’s among brain disorders, may have met its match in an innovative new treatment. Scientists have developed microscopic particles that, when combined with light therapy, successfully reversed Parkinson’s symptoms in mice without requiring brain surgery.
People with Parkinson’s gradually lose brain cells that produce dopamine, a crucial chemical messenger that helps control movement. This loss leads to the disease’s telltale symptoms: tremors, stiff movements, and balance problems.
Current treatments either replace dopamine through medication or use deep brain stimulation via surgically implanted brain electrodes to stimulate remaining healthy cells. These electrodes connect to a device (similar to a pacemaker) placed under the skin in the chest, which sends electrical pulses to control abnormal brain activity.
While these approaches help manage symptoms, they come with significant drawbacks. Medications can lose effectiveness over time, and brain surgery requires permanent hardware implantation and carries risks including cognitive problems and depression.
Scientists at China’s National Center for Nanoscience and Technology, led by Professor Chunying Chen, have created a potential alternative using specially designed nanoparticles — particles so tiny that thousands could fit on the width of a human hair. Their system, nicknamed “ATB NPs,” combines three key components:
- A gold shell that responds to infrared light by generating gentle heat
- A targeting molecule that helps the particles attach to specific brain cells
- A therapeutic protein that breaks down harmful protein clumps that accumulate in Parkinson’s
Think of it as a precision heating system for the brain. When doctors inject these nanoparticles into a specific brain region and shine infrared light (which can pass through skull and tissue) from outside the head, the particles warm up just enough to activate natural heat-sensitive switches on brain cells. This activation helps restore normal cell function while also triggering the brain’s natural cleanup systems to remove toxic protein buildup.
In laboratory tests, mice with Parkinson’s-like symptoms received a single injection of the nanoparticles followed by weekly light therapy sessions for five weeks. The results were remarkable. Treated mice moved almost as well as healthy mice in various physical tests. When researchers examined their brain tissue, they found that previously damaged cells had recovered and toxic protein clusters had diminished.
This approach solves several problems with current treatments. Unlike deep brain stimulation, it doesn’t require permanent hardware in the brain. It also works with the brain’s existing machinery rather than introducing foreign genetic material. The infrared light can be precisely aimed at specific brain regions, allowing for targeted treatment without affecting surrounding areas.
Safety tests showed promising results. The nanoparticles stayed where they were supposed to for at least eight weeks without causing noticeable side effects. This suggests that occasional light treatments might be enough to maintain improvements.
While the research, published in Science Advances, represents an early step toward a potential treatment, it also opens up new possibilities for other neurodegenerative conditions. As one scientist noted in the paper’s discussion, this “wireless” deep brain stimulation therapeutic approach may revolutionize how we treat brain diseases that involve protein aggregation and neuronal dysfunction.
Paper Summary
Methodology
The researchers first developed and characterized their nanoparticle system in laboratory tests, confirming its ability to target dopamine neurons and respond to near-infrared light. They then conducted extensive cell culture experiments before moving to animal studies. In the mouse experiments, they induced Parkinson’s-like conditions using established methods and administered the nanoparticle treatment through precise injections into the substantia nigra. The mice received weekly near-infrared light treatments, and their behavior and brain tissue were analyzed using various standardized tests and imaging techniques.
The researchers specifically chose to target TRPV1 receptors because they are naturally expressed in dopamine neurons, eliminating the need for genetic modification. The near-infrared wavelength of 808 nm was selected for its ability to penetrate brain tissue effectively.
Results
The treatment demonstrated significant therapeutic effects across multiple measures. Treated mice showed improved performance in motor tests, including better balance and coordination. Brain tissue analysis revealed increased survival of dopamine neurons and reduced levels of toxic protein aggregates. The nanoparticles remained stable in the target brain region and showed minimal distribution to other tissues.
The treatment not only improved motor function but also restored the interactive network of dopaminergic neurons and their ability to release dopamine, as demonstrated through various behavioral and cellular analyses.
Limitations
While promising, this study was conducted in mice with artificially induced Parkinson’s-like conditions, which may not fully replicate human disease complexity. The long-term effects beyond eight weeks weren’t evaluated, and the treatment required precise injection into brain tissue, which presents surgical challenges for human application.
Discussion and Takeaways
This research presents a novel approach to treating Parkinson’s disease that combines multiple therapeutic mechanisms in one system. The ability to achieve therapeutic effects without permanent implants or genetic modification represents a significant advance. The demonstrated safety and stability of the nanoparticles suggest potential clinical viability.
This system also represents a significant advance in non-invasive deep brain stimulation technology, combining precise spatial control with strong tissue penetration capabilities. The approach could potentially be adapted for treating other neurodegenerative disorders that involve protein aggregation.
Funding and Disclosures
The research was supported by various Chinese government funding sources, including the National Key Research and Development Program of China and the National Natural Science Foundation of China. The authors declared no competing interests.
Publication Information
Published in Science Advances on January 15, 2025 Title: “A nanoparticle-based wireless deep brain stimulation system that reverses Parkinson’s disease” Authors: Junguang Wu, Xuejing Cui, Lin Bao, Guanyu Liu, Xiaoyu Wang, Chunying Chen DOI: 10.1126/sciadv.ado4927







