Lab mouse unrelated to study. (© filin174 - stock.adobe.com)
DALLAS — Roughly 5.4 million Americans are currently living with paralysis, of which 27.3% are due to spinal injuries. According to a recent study, movement can be restored to mice paralyzed from spinal injuries via genetic engineering. Scientists say this breakthrough could ultimately help paraplegic and quadriplegic patients walk again.
Around 2,500 people per year are left with life changing paralysis, usually as a result of car or sports accidents, violence and falls. After initial damage, sticky scar tissue prevents repair by acting like glue, leading to paralysis below the site.
Scientists at the University of Texas Southwestern discovered a protein in the body that makes regeneration possible. The chemical, known as SOX2, stops scar formation and fuels nerve cells. It is produced by cells in the central nervous system called NG2 glia.
According to study leader Chun-Li Zhang, PhD, this opens the door to a safe and effective therapy that boosts SOX2 in humans. This would help heal spinal injuries by making new neurons and reducing the scar tissue.
“The field of spinal cord injury has extensively researched trying to heal the damage with stem cells that produce new neurons. However, what we are proposing here is that we may not need to transplant cells from the outside. By encouraging NG2 glia to make more SOX2, the body can make its own new neurons, rebuilding from within,” says Zhang, a professor of molecular biology at the university, in a statement.
The findings offer hope for the hundreds of thousands of people worldwide who suffer a spinal cord injury each year. Cells in some tissues and organs proliferate after damage as part of the healing process. This does not apply to the spinal cord – a key roadblock to recovery, Prof Zhang explains.
A promising breakthrough for spinal cord injury patients suffering from paralysis
The spinal cord is the key junction for the constant stream of electrical signals between the brain and the rest of the body. Once harmed, injuries tend to be permanent because nerve cells cannot regenerate themselves. Communication is then halted, leading to paralysis, loss of sensation and sometimes life threatening consequences such as an inability to control breathing or heart rate.
The brain has a limited capacity to fix itself using progenitor cells that can turn into a specific type, like stem cells. Professor Zhang and colleagues looked for cells that might play a similar role in the spinal cord, and identified NG2 glia in the lab rodents.
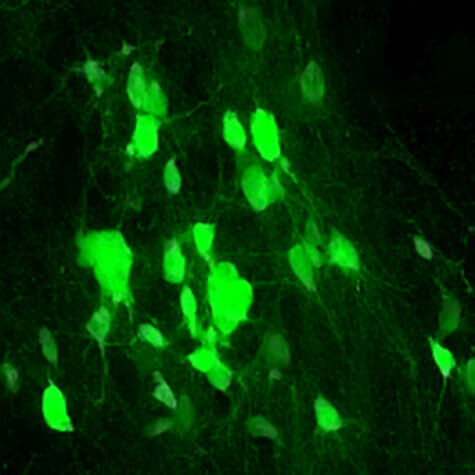
In a series of experiments, they manipulated their DNA to make the cells overproduce SOX2. After their spinal cords were broken, the mice produced tens of thousands of new mature nerve cells. Analysis showed they integrated into the injured area, making connections with existing neurons that are necessary to relay signals between the brain and body.
“Even more promising is that this genetic engineering led to functional improvements. Animals engineered to overproduce SOX2 in their NG2 glia performed markedly better on motor skills weeks after spinal cord injury compared with those that made normal SOX2 amounts,” says Zhang. “The reasons for this improved performance seemed to be multifold. These animals had new neurons that appeared to take over for those damaged during injury. They also had far less scar tissue at the injury site that could hinder recovery.”
A complete recovery from a spinal cord injury would have far reaching social and economic benefits for millions of people.
Findings are published in the journal Cell Stem Cell.
SWNS writer Mark Waghorn contributed to this report.







