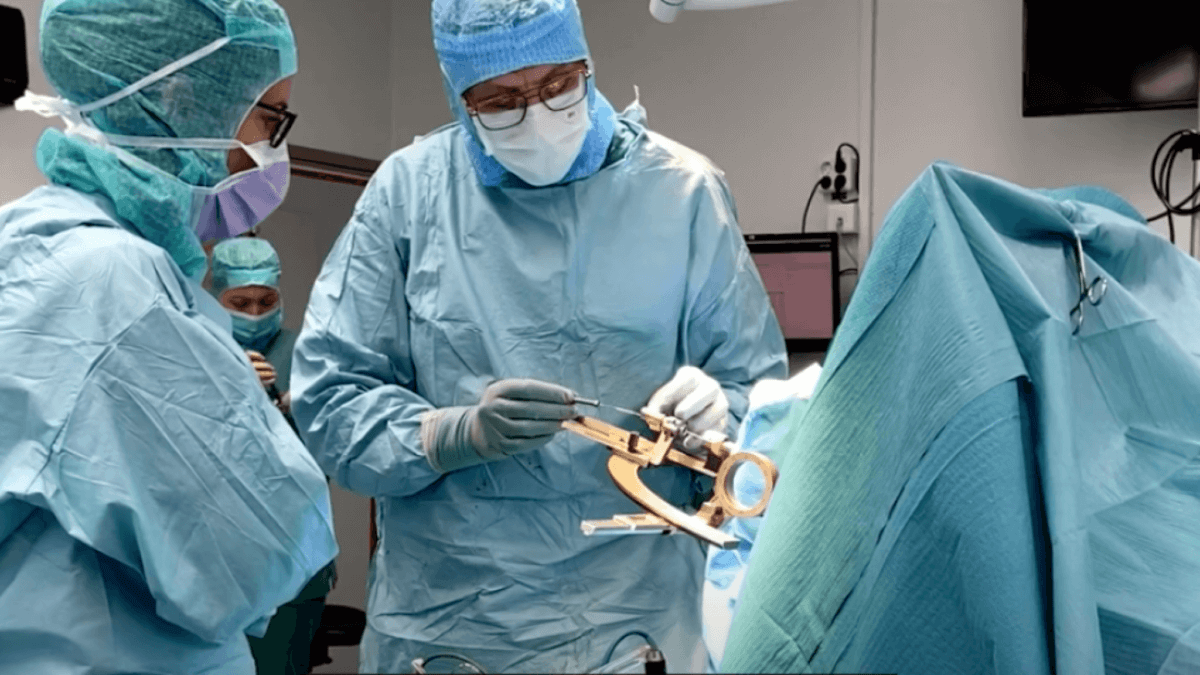
The milestone transplant was performed at Skåne University Hospital in February (Credit: Lund University)
LUND, Sweden — Stem cell transplants may finally provide patients with Parkinson’s disease with hope of a cure. Doctors at Skåne University Hospital successfully transferred a stem cell-derived nerve cell to a Parkinson’s patient for the first time ever in February 2023. It represents a major breakthrough in the treatment of the neurological disorder, according to researchers at Lund University, who developed the procedure.
More specifically, scientists generate the transplantation product using embryonic stem cells. They serve as a replacement for the dopamine nerve cells those with Parkinson’s typically lose as the disease progresses. The initial patient is just the first of eight with Parkinson’s disease who will receive the transplant.
“This is an important milestone on the road towards a cell therapy that can be used to treat patients with Parkinson’s disease. The transplantation has been completed as planned, and the correct location of the cell implant has been confirmed by a magnetic resonance imaging. Any potential effects of the STEM PD-product may take several years. The patient has been discharged from the hospital and evaluations will be conducted according to the study protocol,” says Gesine Paul-Visse, principal investigator for the STEM-PD clinical trial, consultant neurologist at Skåne University Hospital, and adjunct professor at Lund University, in a media release.
Roughly eight million people live with Parkinson’s disease worldwide. The main characteristic of the disease is a loss of dopamine nerve cells deep in the brain, resulting in issues with movement control — such as tremors in the hands. Current standard treatments for Parkinson’s disease usually include meds that replace the lost dopamine, but over time, these medications often end up becoming less and less effective and may cause harmful side-effects.
Currently, there are no treatments capable of repairing the damaged structures within the brain or replacing the lost nerve cells.
Now, the STEM-PD trial is testing a new investigational therapy developed to replace the lost dopamine cells with healthy cells created via stem cells. Using the Swedish Medical Products Agency’s quality standards, researchers subjected their cell product to rigorous pre-clinical tests. After transplantation, scientists expect the cells to mature into new and healthy dopamine-producing nerve cells in the mind.
“With this trial, we hope to demonstrate that the cell product works as expected in patients. Over time, this creates the opportunity to help many more people with Parkinson’s in the future.” adds Lund University professor Malin Parmar.
Parmar leads the STEM-PD team in close collaboration with their colleagues at Skåne University Hospital, Cambridge University, Cambridge University Hospitals NHS Foundation Trust, and Imperial College London.
“Further studies are required to move STEM-PD from this first in human trial all the way to a global treatment, and we have therefore worked in close collaboration with the pharmaceutical company Novo Nordisk A/S. Their input to the study, as well as operational and regulatory guidance, have been fundamentally important to initiate this first in human study and we look forward to future collaborations.”
All in all, eight patients from Sweden and the United Kingdom will undergo this transplantation at Skåne University Hospital, which just so happens to have a long tradition of this type of surgery. The surgical instrument surgeons use in the current trial was originally developed by the hospital for cell transplantations during the 1980s. Back then, stem cells weren’t readily available, so neurosurgeons transplanted nerve cells taken from fetal tissue.
“The brain region that the cells are transplanted into in this trial can be as narrow as four millimeters. The surgical instrument has a very high level of precision, and we are greatly helped by modern imaging techniques” says consultant neurosurgeon Hjálmar Bjartmarz, who actually carried out the transplantation surgery.
All patients in this trial having been living with Parkinson’s for at least a decade. The team considered each of them to be at a moderate stage in their disease progression. The researchers will continue following these patients closely, assessing cell survival rates and potential effects over the coming years.







