(© unlimit3d - stock.adobe.com)
VIENNA, Austria — What if you could press pause on the miracle of life? It sounds like science fiction, but researchers have uncovered evidence that human embryos might have a secret “snooze button.” This incredible finding, detailed in a new study, could potentially revolutionize the future of fertility treatments.
Published in the journal Cell, the study reveals that our embryos may possess a dormancy capability similar to that observed in many other mammals, opening up new possibilities for reproductive medicine and stem cell research.
The ability to enter a state of suspended animation, known as diapause, is a common feature in over 130 mammalian species. This evolutionary trick allows animals to delay the implantation of their embryos in response to unfavorable environmental conditions, effectively putting development on hold until circumstances improve. While this phenomenon has been well-documented in animals like mice, kangaroos, and even bears, it has never been conclusively observed in humans – until now.
In the study, a team of international scientists, led by researchers from the Max Planck Institute for Molecular Genetics in Germany and the Institute of Molecular Biotechnology in Austria, demonstrated that human embryos and stem cells can be coaxed into this diapause-like state. This dormant condition is characterized by slowed growth, reduced energy consumption, and a remarkable ability to resume normal development when the right signals are given.
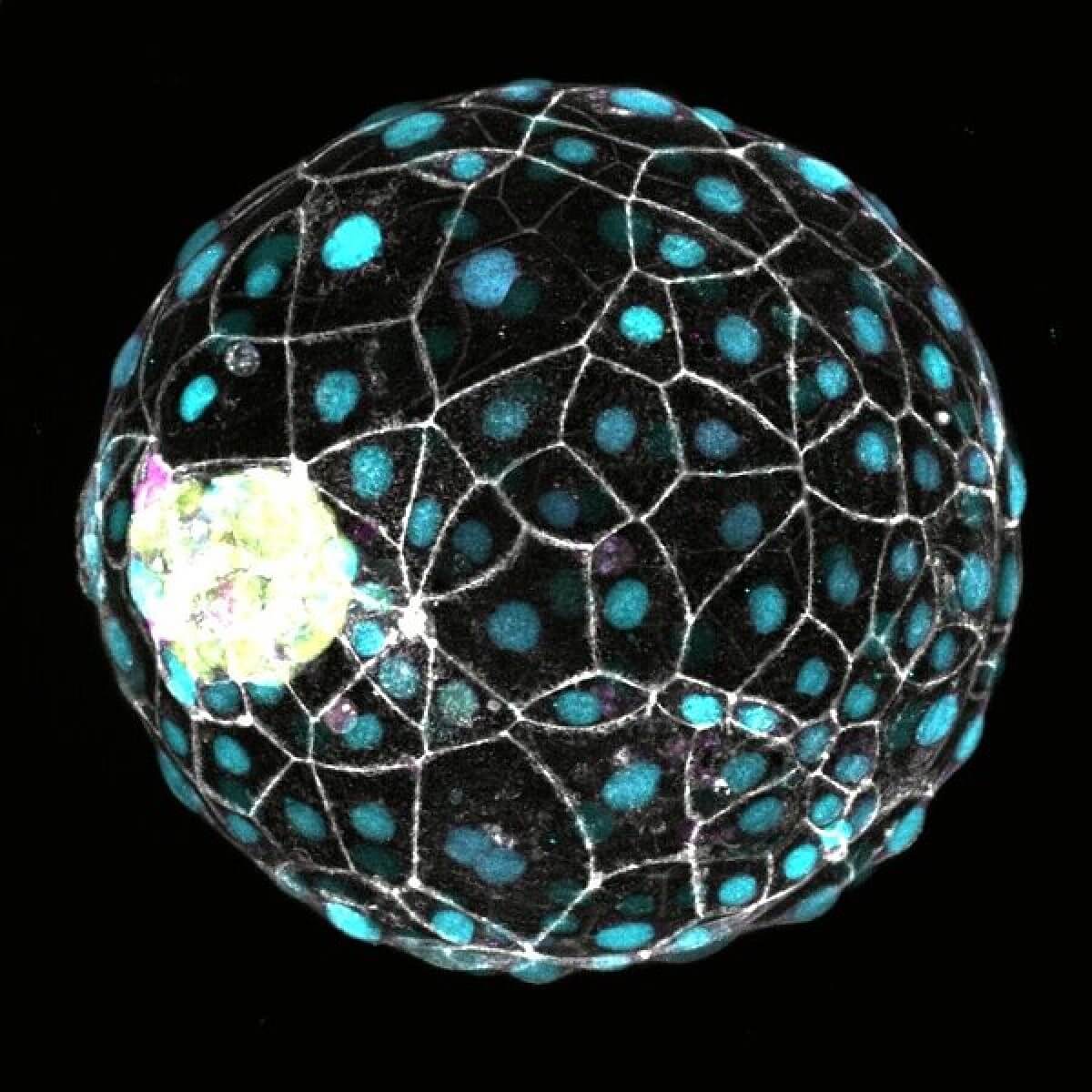
The key to inducing this embryonic pause lies in manipulating a cellular pathway known as mTOR (mechanistic target of rapamycin). By inhibiting mTOR activity, the researchers were able to dramatically slow down the development of human “blastoids” – lab-grown structures that closely mimic early-stage embryos. These blastoids, created using human pluripotent stem cells, provide an ethical alternative to studying actual human embryos while still offering valuable insights into early development.
When treated with mTOR inhibitors, the blastoids entered a state of dormancy that could be maintained for up to eight days. During this time, cell division slowed to a crawl, and the embryo-like structures showed reduced metabolic activity. Remarkably, when the inhibitors were removed, the blastoids “woke up” and continued their development as if nothing had happened.
“When we treated human stem cells and blastoids with an mTOR inhibitor we observed a developmental delay, which means that human cells can deploy the molecular machinery to elicit a diapause-like response,” explains Aydan Bulut-Karslioglu, one of the study’s lead researchers, in a statement.
This ability to pause and resume development could have profound implications for fertility treatments. Currently, the timing of embryo implantation is a critical factor in the success of in vitro fertilization (IVF). The discovery of a way to temporarily halt embryonic development could give doctors more flexibility in timing implantation, potentially increasing the chances of successful pregnancies.
“On the one hand, undergoing faster development is known to increase the success rate of in vitro fertilization (IVF), and enhancing mTOR activity could achieve this,” says Nicolas Rivron, another lead researcher on the study. “On the other hand, triggering a dormant state during an IVF procedure could provide a larger time window to assess embryo health and to synchronize it with the mother for better implantation inside the uterus.”
Moreover, the study sheds light on the fundamental biology of early human development. By comparing the dormant human blastoids to mouse embryos in diapause, the researchers identified both similarities and differences in how various species handle this paused state. This comparative approach provides new insights into the evolutionary conservation of developmental processes across mammals.
“This potential may be a vestige of the evolutionary process that we no longer make use of,” Rivron notes. “Although we have lost the ability to naturally enter dormancy, these experiments suggest that we have nevertheless retained this inner ability and could eventually unleash it.”
The research also has implications beyond reproductive medicine. The ability to induce a dormant state in stem cells could lead to new methods for preserving and transporting these valuable biological resources. Additionally, understanding the mechanisms of cellular dormancy could inform research into cancer, where some tumor cells enter a similar quiescent state to evade treatment.
While the study represents a significant advance in our understanding of human embryonic development, the researchers emphasize that much work remains to be done before these findings can be translated into clinical applications. Ethical considerations and safety concerns must be carefully addressed before any techniques involving the manipulation of human embryos can be implemented in fertility treatments.
Nevertheless, this discovery opens up exciting new avenues for research and potential therapies. By unlocking the secrets of embryonic dormancy, scientists have taken a crucial step towards unraveling the mysteries of human development and may have found a powerful new tool in the quest to help those struggling with infertility.
Paper Summary
Methodology
The researchers used a combination of techniques to study embryonic dormancy. They created “blastoids,” which are lab-grown structures that mimic early-stage embryos, using human pluripotent stem cells. These blastoids were then treated with chemicals that inhibit the mTOR pathway, a key regulator of cell growth and metabolism. The team observed how these treated blastoids behaved over time, measuring things like cell division rates and metabolic activity.
They also performed similar experiments on actual mouse embryos for comparison. Additionally, they used advanced techniques like single-cell RNA sequencing to analyze gene expression patterns in individual cells of the dormant and active blastoids.
Key Results
The study found that inhibiting mTOR activity in human blastoids induced a state of dormancy that could last up to 8 days. During this time, cell division slowed significantly, but the basic structure of the blastoid remained intact. When the mTOR inhibitors were removed, the blastoids resumed normal development. Importantly, the dormant blastoids maintained their ability to form all the cell types needed for early embryonic development, even after the pause. The researchers also observed similarities in gene expression patterns between the dormant human blastoids and mouse embryos in diapause, suggesting a conserved mechanism across species.
Study Limitations
First, the study used blastoids rather than actual human embryos, which may not perfectly replicate all aspects of embryonic development. Second, the induced dormancy could only be maintained for up to 8 days, which is shorter than the diapause period observed in some animals. Additionally, while the blastoids could resume development after dormancy, their long-term developmental potential was not fully assessed. Finally, the safety and ethical implications of manipulating human embryo development in this way need careful consideration before any clinical applications can be pursued.
Discussion & Takeaways
This study provides compelling evidence that human cells possess the molecular machinery to enter a diapause-like state, even though this ability is not naturally used in human reproduction. This discovery has significant implications for both basic science and clinical applications. It offers new insights into the fundamental biology of early human development and the evolutionary conservation of developmental processes across mammals.
From a clinical perspective, the ability to induce temporary dormancy in embryos could lead to improved IVF techniques, allowing for more flexible timing of embryo implantation and potentially increasing success rates. Furthermore, this research opens up new possibilities for stem cell preservation and manipulation, which could have far-reaching impacts in fields such as regenerative medicine and drug discovery.
Funding & Disclosures
The study was supported by various funding sources, including the German Academic Exchange Service, the Swiss National Science Foundation, the Wellcome Trust, the European Research Council, the Austrian Science Fund, the Max Planck Society, and the Humboldt Foundation. Nicolas Rivron, one of the lead researchers, is funded by an ERC Consolidator Grant. The researchers declared no competing interests related to the study.







