
(Photo by Pixabay from Pexels)
STANFORD, Calif. — It’s easy to forget that for many around the world, having clean water is a luxury. Globally, at least two billion people drink water containing disease-causing bacteria. Now, scientists from Stanford University have invented a low-cost, non-toxic, and recyclable powder that can kill thousands of bacteria every second once you expose it to sunlight.
“Waterborne diseases are responsible for 2 million deaths annually, the majority in children under the age of 5,” says study co-lead author Tong Wu, a former postdoctoral scholar of materials science and engineering (MSE) in the Stanford School of Engineering. “We believe that our novel technology will facilitate revolutionary changes in water disinfection and inspire more innovations in this exciting interdisciplinary field.”
Right now, water treatment technologies use chemicals that can produce toxins and UV light, all while taking time to disinfect and requiring electricity to work. This technology developed at Stanford is a simple metallic powder that absorbs both UV and high-energy visible light from the Sun. The powder is made of nano-size flakes of aluminum oxide, molybdenum sulfide, copper, and iron oxide.
“We only used a tiny amount of these materials,” explains senior author Yi Cui, the Fortinet Founders Professor of MSE and of Energy Science & Engineering in the Stanford Doerr School of Sustainability, in a university release. “The materials are low cost and fairly abundant. The key innovation is that, when immersed in water, they all function together.”
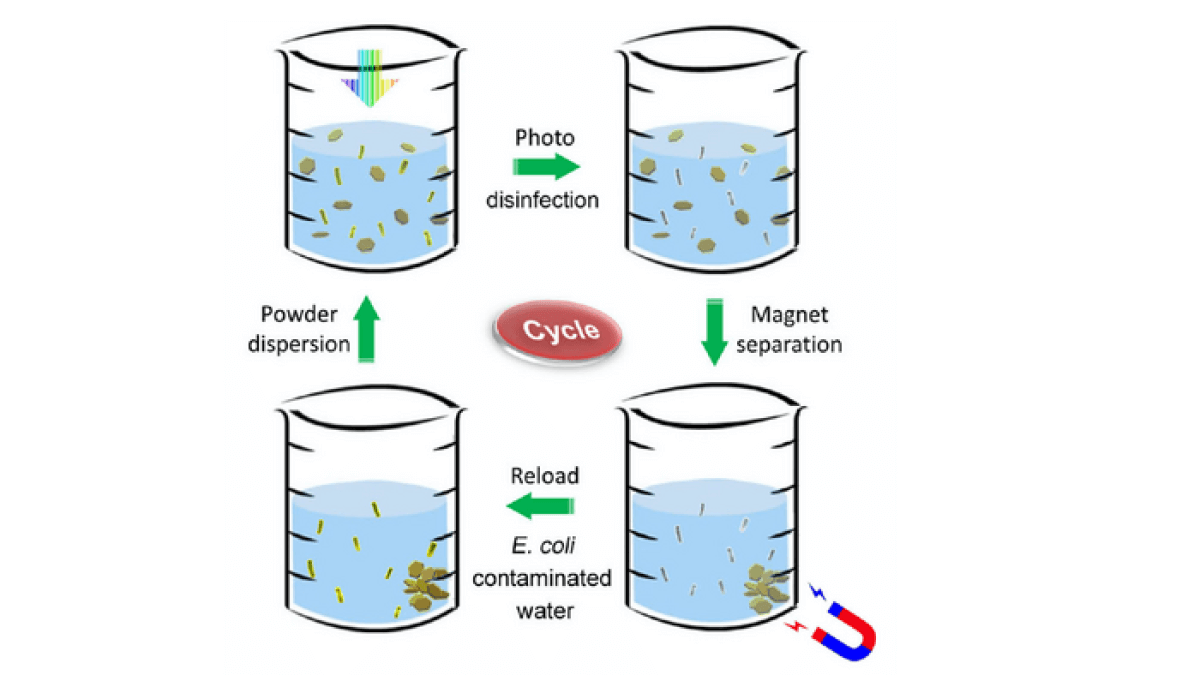
How does the cleaning process work?
Once sunlight particles (photons) are absorbed, the molybdenum sulfide/copper catalyst acts like a semiconductor/metal junction, allowing the photons to displace electrons. The electrons are then able to react with water to create hydrogen peroxide and hydroxyl radicals capable of killing bacteria quickly and efficiently by damaging their cell membranes.
“We stirred the powder into the contaminated water,” says co-lead author Bofei Liu, a former MSE postdoc. “Then we carried out the disinfection test on the Stanford campus in real sunlight, and within 60 seconds no live bacteria were detected.”
“The lifetime of hydrogen peroxide and hydroxy radicals is very short,” adds Cui. “If they don’t immediately find bacteria to oxidize, the chemicals break down into water and oxygen and are discarded within seconds. So you can drink the water right away.”
The iron oxide makes it possible for the powder to be recyclable, allowing the nanoflakes to be removed from the water using a magnet. The team used magnetism to collect the same powder 30 times to treat 30 different samples of contaminated water.
“For hikers and backpackers, I could envision carrying a tiny amount of powder and a small magnet,” says Cui. “During the day you put the powder in water, shake it up a little bit under sunlight and within a minute you have drinkable water. You use the magnet to take out the particles for later use.”
This environmentally-friendly technology could be a game changer for people in developing nations around the world. The Stanford research team looks ahead to testing the powder on other waterborne pathogens, as this study focused on E. coli.
The findings are published in the journal Nature Water.








When and where can this pure water treatment product be available to the general public?