(© dang - stock.adobe.com)
Electrical signals could soon be used to quickly screen patients for diseases
In a nutshell
- Japanese researchers developed a technique using electrical signals to identify aging cells without blood tests or complex lab procedures
- The method can distinguish between young and old cells by measuring their electrical “fingerprints” — aged cells show dramatically different responses to electric fields
- While still experimental and limited to lab conditions, this breakthrough could eventually lead to rapid screening for age-related diseases in clinical settings
TOKYO — Doctors may eventually be able to tell whether your cells are aging prematurely without needles, blood tests, or expensive lab work. Japanese researchers have developed an experimental technique that uses electrical signals to identify senescent cells, which are key players in major health conditions linked to aging.
The breakthrough, published in IEEE Sensors Journal, could eventually transform how we detect and treat age-related diseases, though significant development is still needed.
What Are Senescent Cells and Why Should You Care?
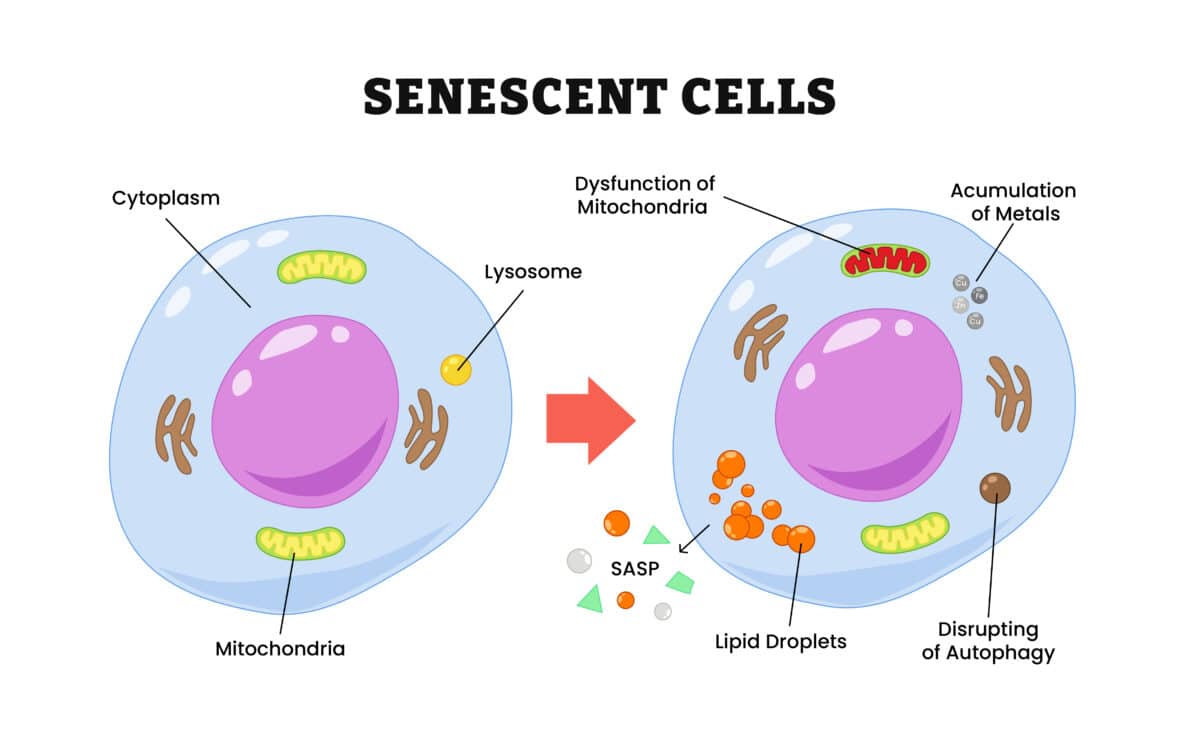
Senescent cells are like cellular retirees: they’ve stopped dividing and growing but haven’t died off. Instead, they linger in your body, often causing inflammation and problems that drive aging and disease. These troublemaker cells accumulate over time and play starring roles in ailments including heart disease, Alzheimer’s, and diabetes.
In heart disease, they build up in blood vessel walls, making arteries stiff and narrow. In the brain, they may worsen Alzheimer’s disease. In diabetes, senescent cells in the pancreas reduce insulin production, making blood sugar harder to control.
Currently, detecting these cells requires complex lab procedures with special stains and microscopes. These processes are time-consuming, expensive, and sometimes unreliable. This new electrical approach developed by scientists at Tokyo Metropolitan University could change all that.
How Scientists Used Electricity to Read Cellular Age
The research team developed frequency-modulated dielectrophoresis (FM-DEP), which sounds complicated but works on a simple principle: cells respond differently to electric fields based on their age and condition.
When researchers place cells in an electric field and vary the frequency, young healthy cells behave one way while aged senescent cells behave another. By watching how cells move and calculating their “crossover frequency” — essentially an electrical fingerprint — scientists can determine whether a cell is young or old.
The team tested this on human skin cells commonly used in aging research. They grew these cells in lab dishes and induced some to become senescent through repeated divisions, mimicking natural aging. Over 2,000 individual cells were analyzed across multiple experiments.
Young Cells vs. Old Cells Show Dramatic Electrical Differences
Young cells typically showed crossover frequencies between 50-150 kHz, while senescent cells displayed much higher and more variable frequencies ranging from 200-500 kHz. The variability in electrical responses increased dramatically in aged cells: up to 27 times more chaotic than their younger counterparts.
Senescent cells were nearly three times larger on average than young cells and showed increased levels of SA-β-galactosidase, a well-known aging marker. The largest, most dramatically aged cells showed the most extreme electrical signatures, with some exceeding 1000 kHz, far beyond healthy cell ranges.
These electrical changes reflect specific molecular transformations in aging cell membranes. As cells become senescent, their outer membranes accumulate more cholesterol and show altered phospholipid ratios, affecting how they respond to electrical fields. “These findings suggest that FM-DEP can differentiate between proliferative and senescence-like phenotypes based on membrane polarization dynamics,” the researchers explained.
Real-World Applications and Current Limitations
The technology could potentially be adapted for medical screening devices, though there’s a long road ahead to move from laboratory conditions to clinical settings. Far from a simple bedside test, the current method requires specialized microfluidic devices, precise electrical equipment, and microscopy analysis.
Researchers could also use electrical measurements to test whether anti-aging treatments actually reverse cellular aging or just mask symptoms.
However, important limitations exist. The study focused only on one type of human cell — skin fibroblasts — grown in artificial lab conditions. Whether the same electrical signatures apply to brain cells, heart cells, or immune cells remains unknown.
All experiments used artificially induced senescence through repeated cell divisions rather than senescence caused by other factors like oxidative stress or DNA damage, which might produce different electrical signatures. As the authors note: “Further studies using diverse cell types and additional senescence markers are needed to assess the broader applicability of this method.”
Despite these constraints, the study represents a significant step toward rapid, label-free detection of cellular aging. The authors concluded that “with continued development — such as improved electrode design and broader biological validation — FM-DEP holds promise for applications in aging research, real-time monitoring of senescence in therapeutic settings, and high-throughput screening of anti-senescence drugs.”
For now, the technique remains a research tool, but it offers a glimpse of a future where detecting cellular aging could be simpler and more accessible than current methods. In a world where aging-related diseases represent an ever-growing health burden, such advances in detection technology are desperately needed.
Paper Summary
Methodology
Researchers used human dermal fibroblasts (TIG-1 cells) grown in laboratory culture dishes. They induced cellular senescence by repeatedly dividing the cells until they stopped growing—a process that mimics natural aging. The cells were categorized as “young” (population doubling level less than 36) or “old” (population doubling level 46-50) based on how many times they had divided. Scientists then used frequency-modulated dielectrophoresis (FM-DEP), applying electrical fields with varying frequencies to individual cells while tracking their movement under a microscope. They calculated each cell’s crossover frequency—the electrical frequency at which the cell’s movement changes direction—as a measure of its electrical properties. The study also measured traditional aging markers including cell size, proliferation rate, and SA-β-galactosidase activity.
Results
The study analyzed over 2,000 individual cells across multiple experiments. Young cells showed crossover frequencies between 50-150 kHz, while senescent cells displayed frequencies of 200-500 kHz with dramatically increased variability (20-27 times more variable than young cells). Senescent cells were approximately 2.8 times larger than young cells on average and showed 88% positivity for SA-β-galactosidase compared to 35% in young cells. The electrical measurements successfully distinguished between young and aged cell populations with statistical significance, and the most extremely aged cells showed crossover frequencies exceeding 1000 kHz.
Limitations
The study was limited to a single human cell type (dermal fibroblasts) grown in artificial laboratory conditions, which may not represent how cells behave in living human bodies. The research only examined senescence induced through repeated cell divisions, not other causes of cellular aging like oxidative stress or DNA damage. The findings need validation across diverse cell types and senescence models before the technique can be considered broadly applicable.
Funding and Disclosures
This research was supported by JSPS KAKENHI grants JP23K28453 and 23KK0260. The authors declared no competing interests. The work was conducted under a Creative Commons Attribution 4.0 License.
Publication Information
The study “Label-free Detection of Senescence-like State in Human Dermal Fibroblasts via Frequency-Modulated Dielectrophoresis” was published in IEEE Sensors Journal on June 11, 2025. The research was led by Ippei Yagi and colleagues from Tokyo Metropolitan University, with additional collaborators from Toyo University, Tokyo Metropolitan Institute of Gerontology, Nanjing Tech University, and National University of Singapore. DOI: 10.1109/JSEN.2025.3576789.







