
(ID 334255041 © Yuri Arcurs | Dreamstime.com)
In A Nutshell
- Researchers identified CGRP-producing neurons in the thalamus that connect pain signals with emotional threat responses.
- These neurons can create fear memories even without actual pain — acting like a built-in brain “alarm system.”
- Silencing these neurons reduced both pain sensitivity and anxiety-like behavior in mice.
- The discovery may shed light on chronic pain disorders and why physical pain so often overlaps with emotional distress.
SAN DIEGO — When someone suffers a painful injury, the physical sensation is just the beginning. The emotional memory of that pain — the fear, anxiety, and dread associated with the experience — can linger long after the wound heals. New research has identified a specific group of brain cells that acts like an internal alarm system, connecting physical pain to emotional distress and threat memories.
The discovery could explain why some people develop chronic pain conditions or become hypersensitive to threats after experiencing trauma. Published in the Proceedings of the National Academy of Sciences, the research reveals how the brain processes not just the sensation of pain, but its emotional meaning.
Scientists at the Salk Institute for Biological Studies focused on neurons that produce a protein called CGRP (calcitonin gene-related peptide) in a brain region called the thalamus. These CGRP neurons function as a central hub, receiving pain signals from the spinal cord and transmitting them to emotional processing centers in the brain.
As the researchers explain: “Pain is both a sensory and emotional experience caused by various harmful stimuli.” Remarkably, these same neurons respond to both physical pain and general threats, indicating the brain uses a unified system to process all forms of danger.

Mapping the Brain’s Pain Highway
The research team used advanced tracing techniques to map the precise connections between the spinal cord and brain. They discovered that CGRP neurons in the thalamus receive direct input from the spinal cord—specifically from the dorsal horn, where pain signals first enter the central nervous system.
To study how these neurons respond to different types of pain, researchers used a technique called fiber photometry, which works like a tiny flashlight that can measure brain cell activity in real time. They applied various painful stimuli to laboratory mice, including mechanical pressure, heat, and chemical irritants that cause inflammation.
CGRP neurons became more active as pain intensity increased. When researchers applied 50 grams of pressure to a mouse’s paw, the neurons showed minimal response. But at 300 grams of pressure, enough to cause significant discomfort, the neurons fired vigorously.
Temperature experiments yielded similar results. At mildly warm temperatures around 77 degrees Fahrenheit, the neurons barely responded. But as heat increased to 131 degrees Fahrenheit, the CGRP neurons became highly active, signaling intense pain.
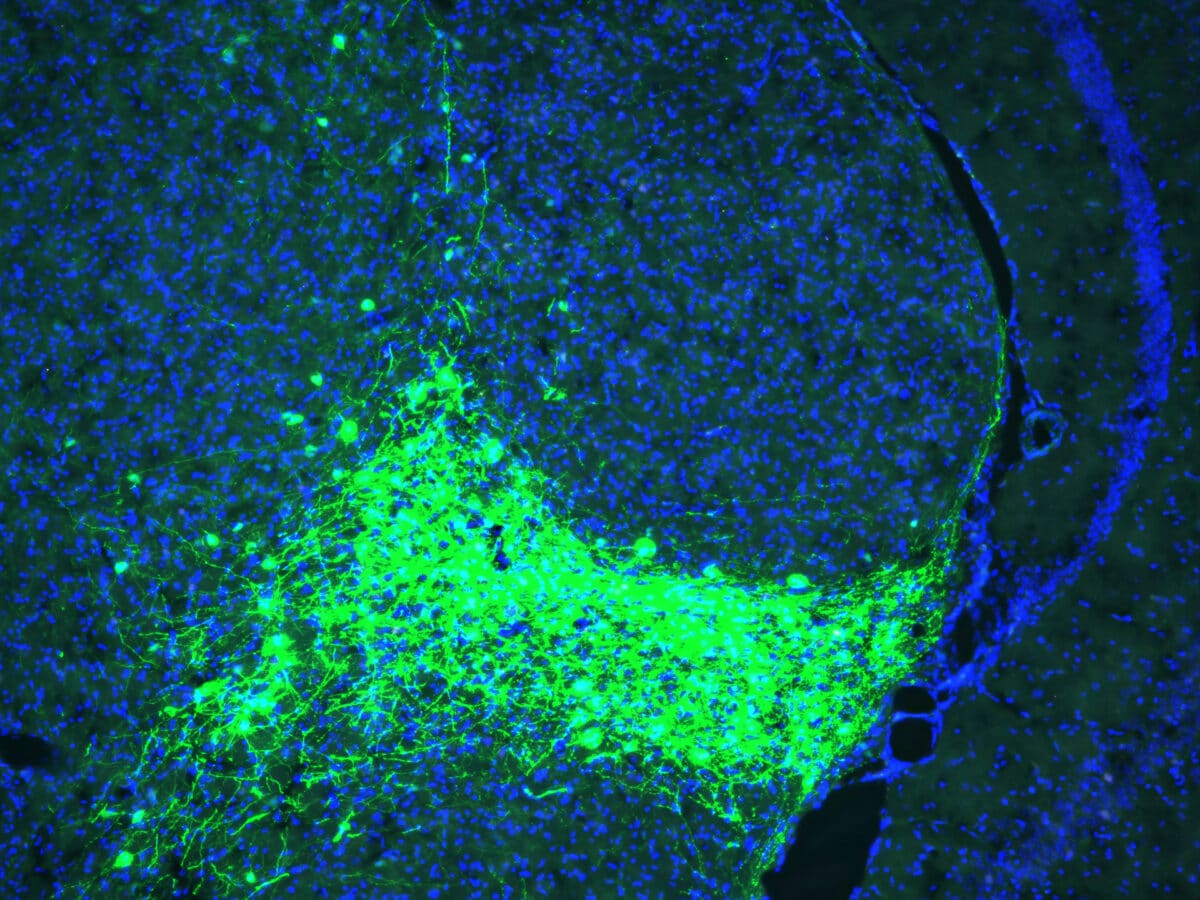
Creating Fear Without Pain
Perhaps most intriguingly, researchers found that artificially activating these CGRP neurons could create threat memories even without any actual painful experience. Using optogenetics, which allows scientists to control specific neurons with pulses of light, they stimulated CGRP neurons while exposing mice to harmless tones.
Later, when the mice heard those same tones, they exhibited fear responses as if they had experienced real trauma. This demonstrates that CGRP neurons don’t just transmit pain signals; they actively create the emotional associations that turn physical pain into lasting psychological distress.
When researchers silenced these neurons using genetic techniques, mice showed reduced pain responses across multiple tests. Animals with silenced CGRP neurons were less sensitive to mechanical pressure, heat, and inflammatory pain. They also displayed less anxiety-like behavior and reduced fear responses in conditioning experiments.
Potential Impact on Human Pain Disorders
The research has potential implications for understanding human pain conditions. Researchers discovered that CGRP neurons express genes linked to several pain-related disorders, including those associated with congenital insensitivity to pain and migraine headaches.
People with congenital insensitivity to pain cannot feel physical discomfort, but they also struggle to recognize general threats, a combination that often leads to shortened lifespans due to repeated injuries. The research suggests this might occur because mutations in pain-processing genes disrupt the same CGRP neurons that handle both pain and threat detection.
Individuals with chronic pain conditions like migraines and fibromyalgia often experience hypersensitivity to normal sensory stimuli. The study found that genes linked to migraines are highly active in CGRP neurons, pointing to these cells’ role in pain hypersensitivity.
CGRP-targeting medications are already approved for migraine prevention, and this research suggests they might be effective for broader applications involving pain and threat processing. The discovery also helps explain why chronic pain often co-occurs with anxiety disorders, post-traumatic stress, and depression.
The researchers conclude: “Together with previous research, our study suggests the presence of a unified threat perception system that integrates multimodal interoceptive and exteroceptive aversive sensory stimuli.”
While the study involved experiments on laboratory mice, the fundamental brain structures and chemical pathways studied are highly conserved across mammalian species, suggesting the findings likely translate to human physiology. Future research could explore whether targeting CGRP neurons might help treat conditions ranging from chronic pain to anxiety disorders and PTSD.
Rather than viewing physical pain and emotional distress as separate phenomena, the discovery of CGRP neurons as a central alarm system shows they’re intimately connected two sides of the same evolutionary coin designed to keep organisms alive and safe from harm.
Paper Summary
Methodology
The researchers used genetically modified mice to study CGRP-producing neurons in two brain regions: the thalamus and parabrachial nucleus. They employed several advanced techniques including viral tracing to map neural connections, fiber photometry to measure real-time brain activity, optogenetics to artificially activate or silence specific neurons, and transcriptomic profiling to identify which genes are active in these cells. Pain responses were tested using mechanical pressure (0-300 grams), temperature stimuli (77-131°F), and inflammatory agents like formalin. Behavioral tests assessed pain sensitivity, anxiety, and fear memory formation.
Results
CGRP neurons in the thalamus receive direct input from spinal cord pain centers and respond to painful stimuli in an intensity-dependent manner. Silencing these neurons reduced pain responses, anxiety, and fear conditioning, while artificially activating them created threat memories without actual pain. CGRP neurons in two different brain regions showed distinct response patterns—thalamic neurons preferred mechanical stimuli while parabrachial neurons were more sensitive to temperature. Both neuron types expressed genes linked to human pain disorders including migraine and congenital pain insensitivity.
Limitations
The study was conducted entirely in laboratory mice, so the findings need validation in humans. Experiments used anesthetized animals for some measurements, which may not fully reflect natural pain processing. The research focused on specific CGRP neuron populations and didn’t examine all possible pain pathways in the brain. Long-term effects of manipulating these neurons weren’t assessed.
Funding and Disclosures
The research was supported by grants from the National Institute of Mental Health (1R01MH116203) and the Simons Foundation Autism Research Initiative Bridge to Independence award (#388708). The authors declared no competing interests.
Publication Information
“Thalamic CGRP neurons define a spinothalamic pathway for affective pain,” by Sukjae J. Kang and colleagues, published in Proceedings of the National Academy of Sciences, 2025, Volume 122, DOI: doi.org/10.1073/pnas.2505889122.








“The discovery also helps explain why chronic pain often co-occurs with anxiety disorders, post-traumatic stress, and depression.”
Hopefully governments won’t pull support from people who experience debilitating pain due this this phenomenon before a way to effectively and efficiently manage, maybe even cure, chronic pain is found and then applied.