(Credit: VideoBCN/Shutterstock)
NEW YORK — For the millions of Americans who suffer from shingles each year, the threat of vision loss has long been a looming concern. Now, groundbreaking research offers a glimmer of hope: a simple, extended course of antiviral medication may significantly reduce the risk of eye damage and chronic pain associated with this painful condition.
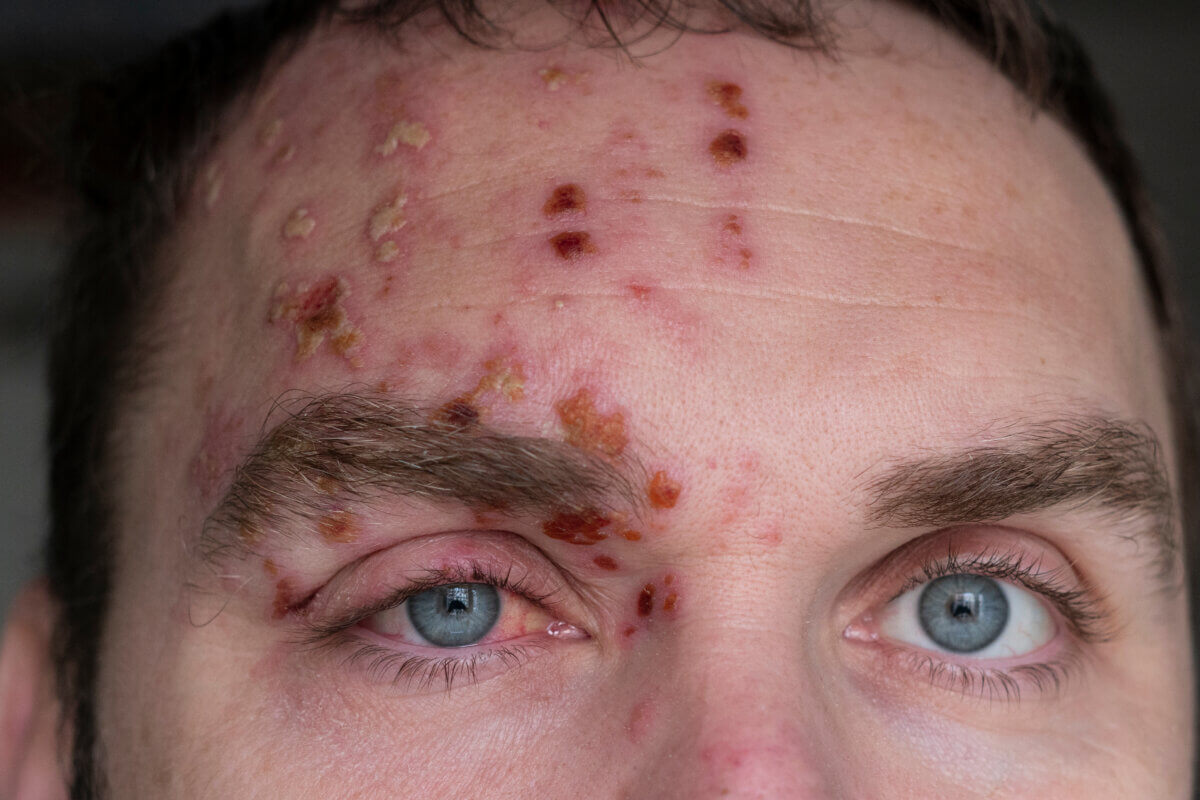
Shingles, caused by the reactivation of the chickenpox virus, affects more than a million people in the United States annually. For about 100,000 of these individuals, the virus targets the eye in a condition known as herpes zoster ophthalmicus (HZO). This can lead to severe complications, including inflammation of the cornea and iris, and even legal blindness in some cases.
Until now, treatment options for preventing long-term eye damage from HZO have been limited. However, a new study, presented at the American Academy of Ophthalmology’s annual meeting in Chicago, suggests that a year-long course of the antiviral drug valacyclovir could be a game-changer.
“Our results support changes in clinical practice, with suppressive valacyclovir recommended to reduce new, worsening, and repeated episodes of eye disease, as well as need for neuropathic pain medication in HZO patients and in those with shingles-related pain,” explains Dr. Elisabeth Cohen, a professor of ophthalmology at NYU Grossman School of Medicine and the study’s principal investigator, in a media release.
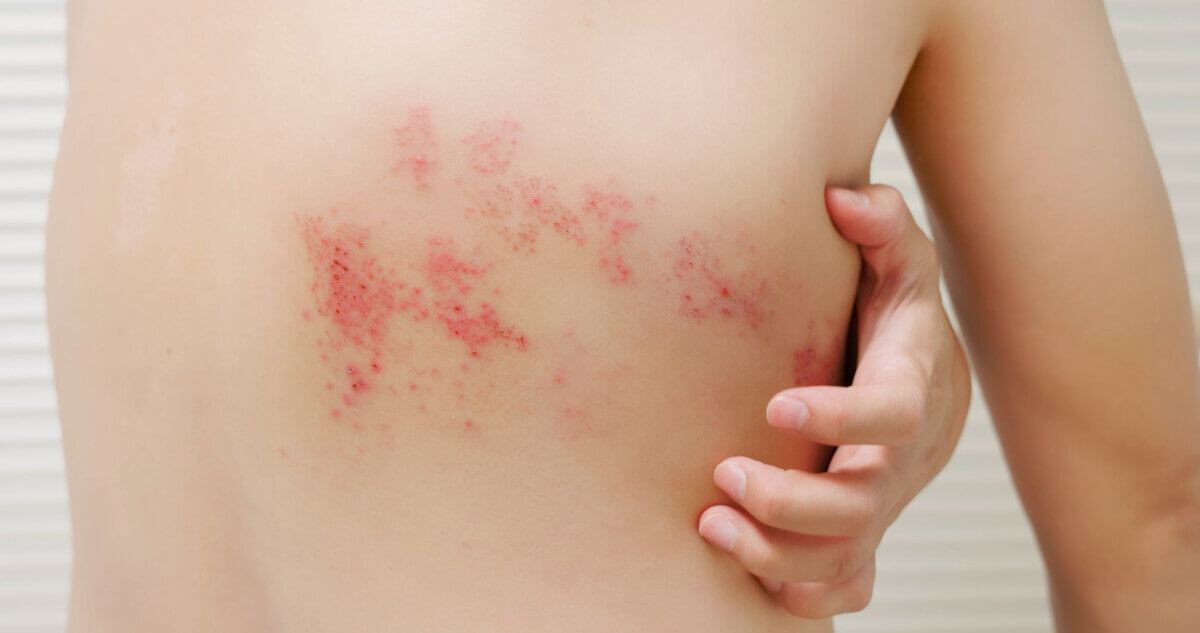
The study, known as the Zoster Eye Disease Study (ZEDS), involved 527 participants across 95 medical centers in the United States, Canada, and New Zealand. Half of the participants received a daily dose of valacyclovir for a year, while the other half received a placebo.
The results were striking. Those treated with valacyclovir saw a 26% reduction in their risk of developing new or worsening eye disease 18 months after starting treatment. Moreover, they were 30% less likely to experience multiple disease flare-ups at one year and 28% less likely at 18 months.
What is valacyclovir?
Valacyclovir is an antiviral medication used to treat various viral infections, primarily those caused by herpes viruses. It’s a prodrug of acyclovir, which means that after ingestion, it’s converted in the body to acyclovir, the active form of the medication. The drug often goes by its brand name Valtrex.
It’s commonly prescribed to patients with herpes simplex virus (HSV) infections, including genital herpes and cold sores, but is also given to people with shingles and chickenpox. It’s generally considered safe and well-tolerated. Like all medications, however, it can have side-effects in some people.
The benefits didn’t stop there. The treatment also showed promise in reducing the chronic nerve pain that often accompanies shingles, particularly in older patients. This is crucial, as current pain medications for this condition have limited effectiveness and can cause troublesome side-effects like dizziness.
“The results of this study provide convincing evidence for using long-term, low-dose antiviral treatment to reduce eye disease in HZO and decrease pain from shingles,” says Dr. Bennie Hau Jeng, chair of the Department of Ophthalmology at the University of Pennsylvania and the study’s co-chair.
For many, this new approach could mean the difference between maintaining clear vision and facing potential blindness. However, both Dr. Cohen and Dr. Jeng stress that the prevention of shingles remains the best strategy. The shingles vaccine, recommended for all adults 50 and older, as well as immunocompromised adults 19 and older, is highly effective at preventing the disease altogether. Yet, alarmingly, only about 12% of people in their 50s have received this crucial vaccination.
As research continues, this study opens new doors for those affected by shingles and its ocular complications. It offers not just a treatment but hope – hope for clearer vision, less pain, and a better quality of life for millions of people around the world.
Paper Summary
Methodology
The ZEDS study was a double-blind, randomized, controlled trial. This means that participants were randomly assigned to either the treatment group (receiving valacyclovir) or the control group (receiving a placebo), and neither the participants nor the researchers knew who was in which group during the study. This design helps eliminate bias and ensures that any differences observed between the groups can be attributed to the treatment itself.
Key Results
The key findings show that long-term valacyclovir treatment reduced the risk of new or worsening eye disease by 26% at 18 months. It also decreased the likelihood of multiple disease flare-ups by 30% at 12 months and 28% at 18 months. Additionally, patients on valacyclovir experienced less pain and required fewer pain medications.
Study Limitations
While the study provides promising results, it’s important to note that it focused on patients with functioning immune systems and kidneys, and those over 18 with a history of HZO. The findings may not apply to all shingles patients, particularly those with compromised immune systems or other health conditions.
Discussion & Takeaways
This research suggests a significant shift in how we approach the long-term management of shingles-related eye disease. The extended use of valacyclovir could potentially become a new standard of care, offering better outcomes for patients at risk of vision loss and chronic pain. However, the study also underscores the importance of prevention through vaccination, which remains the most effective strategy against shingles and its complications.
Funding & Disclosures
The ZEDS study was primarily funded by the National Eye Institute, part of the National Institutes of Health. Additional support came from the National Shingles Foundation and a grant from the organization Research to Prevent Blindness. It’s worth noting that Dr. Jeng disclosed receiving financial compensation in the past from a company that makes shingles vaccines, which is important for transparency in medical research.







