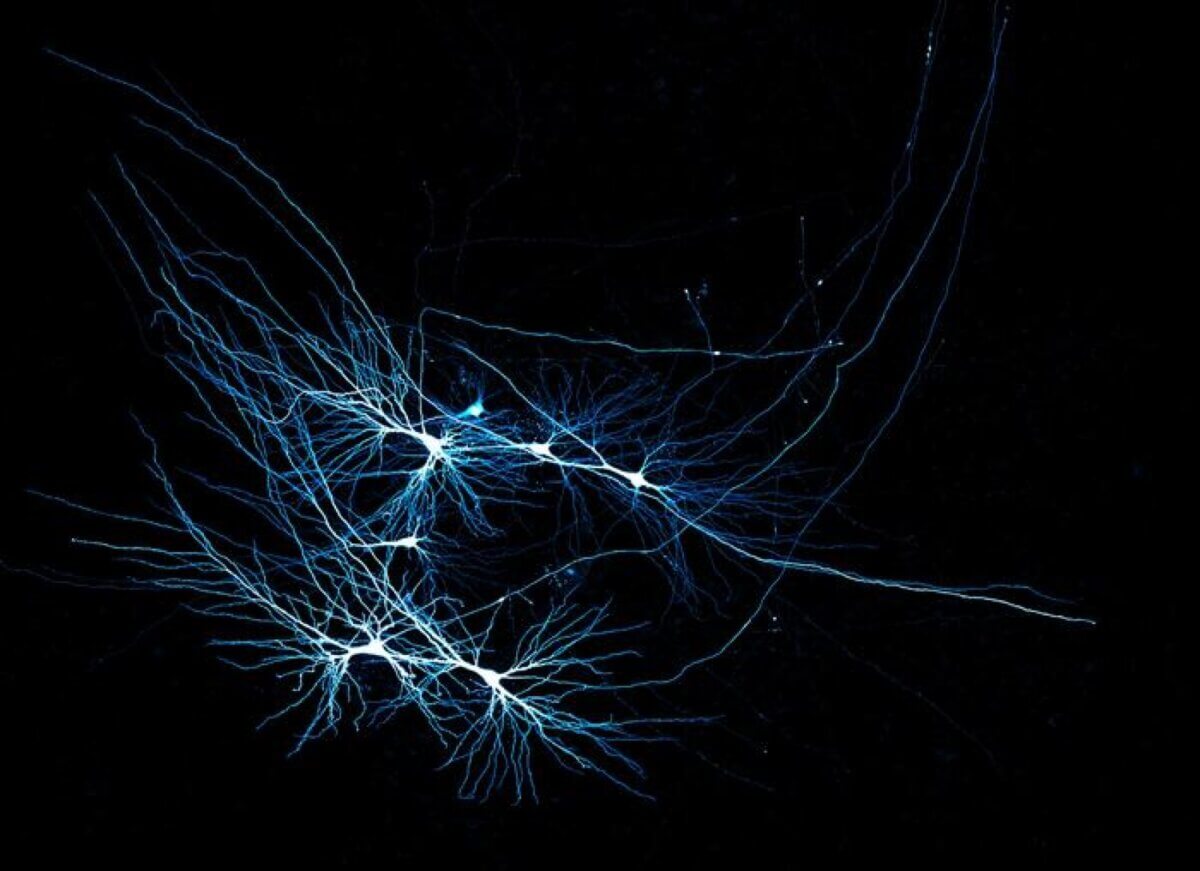
Human CA3 pyramidal neurons recorded in a patient tissue sample. (Credit: © Jake Watson)
KLOSTERNEUBURG, Austria — Imagine listening to your grandparents weave a captivating story about their childhood. How does our brain manage to store and retrieve those vivid memories with such remarkable precision? A new study from the Institute of Science and Technology Austria (ISTA) pulls back the curtain on one of neuroscience’s most fascinating puzzles, revealing what’s really going on inside the brain’s “black box.”
For years, scientists have relied on rodents to understand how our brains work. But what if a mouse brain and a human brain aren’t simply scaled versions of each other? This is exactly what researchers discovered when they took an unprecedented look inside the human hippocampus — the brain’s memory center.
By collaborating with neurosurgeons specializing in epilepsy treatment, the research team gained access to a rare treasure: intact, living human brain tissue from 17 patients undergoing surgery. What they found was nothing short of revolutionary.
“As soon as I started examining the first patient samples, I realized how much we didn’t know about the human hippocampus,” says Jake Watson, a postdoctoral researcher involved in the study, in a media release. “Although this is the best-studied brain region in rodents, it felt like we didn’t know a thing about human physiology.”

Using cutting-edge techniques like multicellular patch-clamp recording and super-resolution microscopy, the researchers discovered that the human brain’s neural connectivity is fundamentally different from what we’ve observed in mice. The human hippocampus, specifically a region called CA3 crucial for memory storage, has sparser connections between neurons. Surprisingly, these connections are also more reliable and precise.
Think of it like comparing a rough sketch to a detailed painting. Where a mouse brain might have numerous, somewhat imprecise neural connections, the human brain appears to have fewer but more targeted pathways. This unique wiring might explain our extraordinary ability to form complex memories and make intricate associations.
“Our work highlights the need to rethink our understanding of the brain from a human perspective. Future research on brain circuitry, even if using rodent model organisms, must be conducted with the human brain in mind,” emphasizes Peter Jonas, the study’s lead researcher and Magdalena Walz Professor for Life Sciences.
The study not only challenges existing neuroscientific models but also opens up exciting new avenues for understanding how we store and retrieve memories. From those cherished moments with grandparents to the complex neural processes that allow us to learn and adapt, our brains continue to surprise and fascinate researchers.
“Looking back, the best day in my career as a physiologist was when the first human tissues arrived in our lab,” Watson recalls.
For science, it was a moment of profound discovery — a small window into the intricate, mysterious landscape of human consciousness.
Paper Summary
Methodology
The researchers studied how human hippocampal CA3 neurons work by analyzing brain tissue from epilepsy surgery patients. They used advanced techniques like multicellular patch-clamp recording and superresolution microscopy to observe the structure and connections of neurons in this region. The team also developed computational models to understand how these networks store memories. They focused on “non-sclerotic” tissue, ensuring they studied healthy-like samples. These experiments revealed how the CA3 circuit connects sparsely but reliably to enhance memory capacity.
Key Results
The study found that the human CA3 region in the brain, crucial for memory, has a unique way of working. Unlike other parts of the brain, CA3 uses sparse but precise connections, meaning fewer neurons connect with each other, but they do so reliably. This setup allows the brain to store and recall memories efficiently without overload. The findings also showed that human CA3 neurons have extended dendrites (branches) and longer-lasting signals compared to rodents, giving humans a more powerful memory network.
Study Limitations
While the study provides significant insights, it was limited by the use of brain tissue from epilepsy patients, which may not fully represent a healthy brain. The sample size was small due to the difficulty in obtaining such tissue. Additionally, the researchers only analyzed local connectivity, leaving broader hippocampal connections unexplored. Future research with completely unaffected tissue and more extensive sampling is needed to validate these findings.
Discussion & Takeaways
The study emphasizes that human CA3 neurons are specialized for efficient memory storage. Sparse connectivity reduces noise in the network, while high reliability ensures accurate signal transmission. These features make human hippocampal memory systems distinct from those in rodents. Importantly, the findings suggest that memory capacity in humans has evolved through expanded neuronal numbers and better connectivity, rather than merely scaling up existing rodent brain systems. This research highlights the importance of studying human-specific brain features for deeper understanding.
Funding & Disclosures
This study was supported by the European Research Council (ERC) under the Horizon 2020 program (advanced grant no. 692692 to P.J. and Marie Skłodowska-Curie Fellowship no. 101026635 to J.F.W.), the Austrian Science Fund (FWF; grant PAT 4178023 to P.J. and grant DK W1232 to M.R.T. and J.G.D.), the Austrian Academy of Sciences (DOC fellowship 26137 to M.R.T.), and a NOMIS-ISTA fellowship (to A.N.-O.).
The authors acknowledge the contributions of patient donors, the NABCA biobank, and postmortem tissue donors, as well as support from ISTA Scientific Services Units and staff at the Medical University of Vienna. M.R.T. and J.G.D. are inventors on a patent application covering expansion microscopy technology, and there are no additional competing interests reported.








I have a very good memory. It’s just short.
Medical researchers ought to collaborate with computer scientists more. I’ve suspected for years that access to memory, both long and short term, is via a ‘hash table’ of some sort. Deterioration of memory is degradation of the biology constructing the hash table, ie the memories may still be there but the route is corrupted. May also explain the ‘life flashing before you” as the tables are traversed entirely under certain circumstances. May also account for the described sparsely of connections. That is the number of ‘collisions’ ie hashing function hashing to same index to the memory, must be kept to a minimum in order to allow fast access. This necessitates storage capacity far exceeding the potential memories stored.
Med
I’m replying to myself. I forgot to mention damaged memory recall as well.
Thank you.
Any way to repair long and short term memory storage damaged by the polio vaccine in 1951?
Can the right neurons be replaced somehow?
Fascinating! And to think that it all started with a Big Bang caused by NOTHING! Not a Big Bang created by a Creator. It’s amazing that NOTHING can start such a process as us and our brains etc.
Nothing comes from nothing