
Patient with forms of aggressive breast cancer often have access to limited treatments options that aren't harmful to their healthy body tissue. (© Pixel-Shot - stock.adobe.com)
BOSTON — A new cancer vaccine uses a unique approach to both destroy brain tumors and keep the disease from returning. Researchers from Brigham and Women’s Hospital say this “dual-action” vaccine turns live cancer cells into potent, cancer-killing agents. Moreover, the vaccine helps a patient’s immune system remember what these cancer fighters “look like” — leading to long-lasting protection.
In advanced experiments with mice, the team found that their genetically-engineered tumor cells produced promising results while fighting off the deadly brain cancer glioblastoma. The average survival length for patients with glioblastoma is about 18 months. Less than five percent of patients live longer than five years after their diagnosis.
“Our team has pursued a simple idea: to take cancer cells and transform them into cancer killers and vaccines,” explains corresponding author Khalid Shah, MS, PhD, director of the Center for Stem Cell and Translational Immunotherapy (CSTI) and the vice chair of research in the Department of Neurosurgery at the Brigham, in a media release. “Using gene engineering, we are repurposing cancer cells to develop a therapeutic that kills tumor cells and stimulates the immune system to both destroy primary tumors and prevent cancer.”
How does the vaccine work?
Cancer vaccines are not a new concept and researchers around the world are currently working on different versions. However, this new study took a very different approach. Instead of using inactive tumor cells, Shah’s team repurposed living cancer cells that had an unusual feature. These particular tumor cells travel long distances across the brain just to return to the site of their fellow cancer cells — just like a homing pigeon.
Shah’s team re-engineered these cells using the gene editing tool CRISPR-Cas9 to release a tumor-killing agent when they returned to the cancer site. Additionally, the team altered these cells to express genetic factors that make them easier for the immune system to identify. Immune cells can then tag and remember the modified cancer fighters — priming the patient’s body for the future and making them less likely to suffer a cancer recurrence.
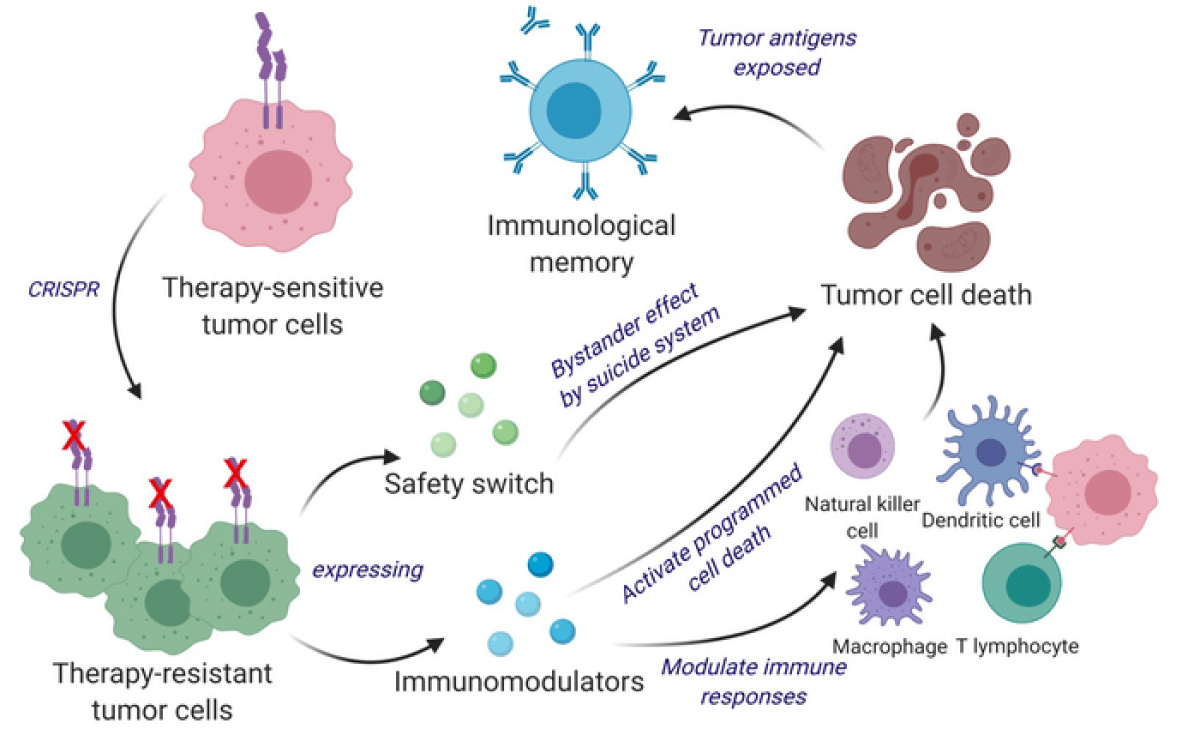
Can this vaccine beat other forms of cancer?
Study authors tested their CRISPR-enhanced and reverse-engineered therapeutic tumor cells (ThTCs) in different mice strains. The mouse samples included bore bone marrow, liver, and thymus cells derived from humans, mimicking the human immune microenvironment.
Results show that the dual-action cell therapy was safe to use and effective in these experiments. Researchers are hopeful this paves the way towards human testing.
Shah also notes that the repurposed tumor cells contain a “two-layered safety switch.” When activated, the switch can eradicate ThTCs if necessary — like a self-destruct switch for cancer cells.
“Throughout all of the work that we do in the Center, even when it is highly technical, we never lose sight of the patient,” Shah concludes. “Our goal is to take an innovative but translatable approach so that we can develop a therapeutic, cancer-killing vaccine that ultimately will have a lasting impact in medicine.”
Shah and his colleagues add that this therapeutic strategy would also work on a wider range of solid tumors. Further investigations into how the vaccine would interact with these tumors is still necessary. Solid tumors include sarcomas (in the bone), carcinomas (in the epithelial tissue of the skin or lining of the internal organs), and lymphomas (in the lymph system).
Shah owns equity in and is a member of the Board of Directors of AMASA Therapeutics, a company developing stem cell-based therapies for cancer.
The findings are published in the journal Science Translational Medicine.







