
Credit: Kateryna Kon on Shutterstock
Virus-built silver appears much more effective against bacteria than commercial silver.
In A Nutshell
- Lab safety tests showed no harm to human kidney cells at concentrations 10 times higher than needed to kill bacteria, though animal and human studies are still needed
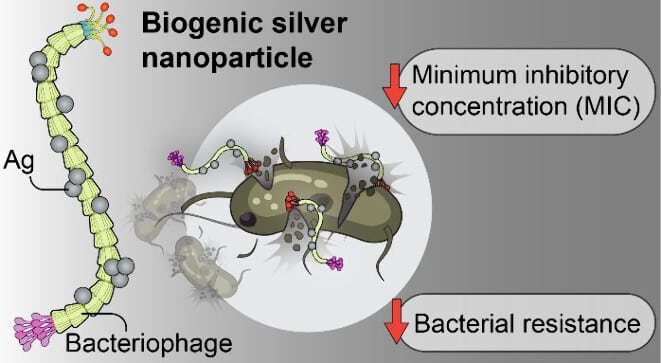
- UCLA researchers used M13 viruses to build silver nanoparticles that killed bacteria at concentrations 30-60 times lower than commercial silver products in lab tests
- Bacteria developed resistance to virus-built nanoparticles more than 10 times slower than conventional versions; tolerance increased only 2-4 fold after 15 rounds versus 24-fold for commercial products
- The particles destroyed more than 50% of bacterial biofilms (the slimy protective fortresses bacteria form on medical devices) and worked against multiple dangerous pathogens including hospital superbugs
Silver has been used to kill bacteria for centuries, but scientists at UCLA have discovered a way to make it work much more effectively. By letting viruses build tiny silver particles, researchers created a material that’s more than 30 times stronger than conventional silver treatments in laboratory tests. Results show that bacteria develop resistance to it much more slowly.
Drug-resistant bacteria caused 2.8 million infections in the United States in 2019, and cases jumped 20% by 2022, according to the CDC. Current silver products work, but they require high doses that raise safety concerns, and bacteria quickly adapt to resist them. These virus-built nanoparticles address both problems, at least in the lab.
Lead researcher Irene Chen and her team at UCLA’s Department of Chemical and Biomolecular Engineering showed that bacteria exposed repeatedly to commercial silver nanoparticles developed substantial resistance after just a few rounds of treatment (the amount needed to kill them increased 24-fold after 15 rounds). The virus-templated versions showed only a 2- to 4-fold increase over the same period, depending on the formulation.
How Viruses Build Better Silver
The researchers enlisted help from M13, a microscopic rod-shaped virus that naturally infects bacteria. The virus is about 7 nanometers wide and roughly 1,000 nanometers long. Its outer shell contains a protein with a negative electrical charge that attracts and converts silver ions into metallic particles.
The process is surprisingly simple. Mix the viruses with silver in water, stir at room temperature, and the viruses do the rest. Depending on how long you stir, be it two hours or a full day, you get different-sized particles. Both versions worked exceptionally well, but the smaller particles proved most potent.
What makes these virus-built particles special isn’t the network structure they form. When researchers tested that theory by mixing regular silver particles with viruses, nothing changed. The real difference lies in the shape of the silver crystals themselves. Virus-built particles expose crystal surfaces that are more chemically active against bacteria than those formed through conventional manufacturing.
Think of it like the blade of a knife: the same metal can be ground to different edge angles, and some cut better than others. The virus creates silver particles with the bacterial equivalent of a razor-sharp edge.
Powerful at Tiny Doses
The new particles stopped bacterial growth at concentrations about 30-60 times lower than commercial silver products tested in the same experiments. They worked against multiple dangerous bacteria, including Pseudomonas aeruginosa, a notorious hospital pathogen that causes pneumonia and bloodstream infections, and Vibrio cholerae, which causes cholera.
Even bacterial biofilms couldn’t resist. Biofilms are slimy protective communities that bacteria form on surfaces like catheters and implants—think of them as bacterial fortresses. These structures make infections incredibly difficult to treat. The virus-built nanoparticles broke down more than half of the biofilm’s protective coating, while commercial versions managed only 10 to 40% reduction.
The effectiveness wasn’t due to the virus infecting bacteria. Tests on bacterial strains that can’t be infected by M13 showed identical results, confirming the silver particles themselves deserve the credit.
Bacteria Adapt Much More Slowly
Perhaps most importantly, bacteria struggled to develop resistance as quickly. The researchers grew bacteria through 15 successive generations, constantly exposing them to sublethal amounts of silver, conditions that typically force rapid evolution of resistance.
Commercial silver nanoparticles failed the test. Resistant bacteria emerged by round 9, requiring eight times more silver to kill. By round 15, bacteria needed 24 times the original concentration. The virus-built versions stayed effective through round 8 with no change. Even after 15 rounds, bacteria had only increased their tolerance by 2- to 4-fold, more than 10 times slower than commercial versions.
Why can’t bacteria adapt as easily? The unusual crystal structure appears to attack through mechanisms bacteria find harder to counter. Known resistance strategies include producing proteins that clump the silver particles together or activating pumps that flush silver out of cells. The virus-built particles may simply overwhelm these defenses more effectively.
Safe for Human Cells in Lab Tests
Safety testing in the laboratory showed no significant harm to a human kidney cell line (HEK293T) at concentrations more than 10 times higher than needed to kill bacteria. Tests with sheep red blood cells showed less than 2% damage across all tested concentrations.
This safety margin could matter if the findings translate to medical use. The new particles work at concentrations below 1 microgram per milliliter—dramatically lower than what’s needed for conventional silver products. That difference could reduce exposure and potential side effects, though animal and human testing would be needed to confirm this.
The findings, published in Langmuir, could eventually expand silver’s medical use beyond topical treatments. Right now, silver products are limited to wound dressings, bandages, and coated catheters. The improved potency and delayed resistance development make these virus-built particles worth investigating for more applications, though the researchers caution that substantial additional testing is required.
The silver nanoparticles did show some tendency to clump together over time in salt solutions, which could affect stability in the body. And while phage production faces manufacturing challenges around consistency and purity, ongoing clinical trials of phage therapies for other purposes suggest these hurdles can be overcome.
For now, the research demonstrates that nature still has lessons to teach us about fighting bacteria. By letting viruses do what they do naturally, scientists created a material that outperforms human engineering by significant margins in laboratory conditions.
Paper Notes
Limitations
The research tested the nanoparticles only against Gram-negative bacteria, one of two major bacterial categories. Activity against Gram-positive species, which include Staphylococcus and Streptococcus, remains unknown. All experiments were conducted in laboratory dishes, not in living animals or humans. The materials showed some clumping over time in salt solutions, which could affect how they behave in the body. The study did not evaluate the nanoparticles in animal models or address potential issues with long-term stability, how they distribute in the body, or long-term toxicity.
Funding and Disclosures
This research was supported by the National Institute of General Medical Sciences of the National Institutes of Health under Grant R35GM148249 and the National Science Foundation BioPACIFIC MIP under Grant DMR-1933487. Author Irene A. Chen is a co-founder of Paralos Bioscience.
Publication Details
Title: Silver Nanoparticles Templated by the M13 Phage Exhibit High Antibacterial Activity against Gram-Negative Pathogens and a Reduced Rate of Bacterial Resistance In Vitro
Authors: Damayanti Bagchi, Aniruddha Adhikari, Katherine McCarthy, Dayeon Kang, Yueyun Chen, and Irene A. Chen | Institution: Department of Chemical and Biomolecular Engineering, Department of Chemistry and Biochemistry, University of California, Los Angeles | Journal: Langmuir | Publication Date: November 27, 2025 | Volume: 41, Pages 32270-32284 | DOI: 10.1021/acs.langmuir.5c03695







