
(Credit: PPStudio on Shutterstock)
Research provides the first-ever high-resolution, cell-resolved map of the human scalp during early aging.
In A Nutshell
- Hair follicle aging starts earlier than you might think: molecular changes linked to inflammation appear in the 30s and 40s, years before visible thinning or graying
- A protein complex called AP-1 shows increased activity in middle-aged hair follicles and may coordinate a network of genes that affect stem cell function
- Cell-to-cell communication weakens in aging scalps, particularly signals between support cells and the cells that grow hair, which may extend rest periods between growth cycles
- The research identifies molecular patterns that could become targets for future treatments, though no therapies were tested in this study
Thinning manes and those first pesky strands of gray hair usually appear in middle age – but the cellular groundwork is laid long before then. According to groundbreaking research mapping human hair follicles at the cellular level, early molecular changes associated with hair aging begin years earlier. Scientists say these changes are detectable in the 30s and 40s, with many tied to chronic low-grade inflammation.
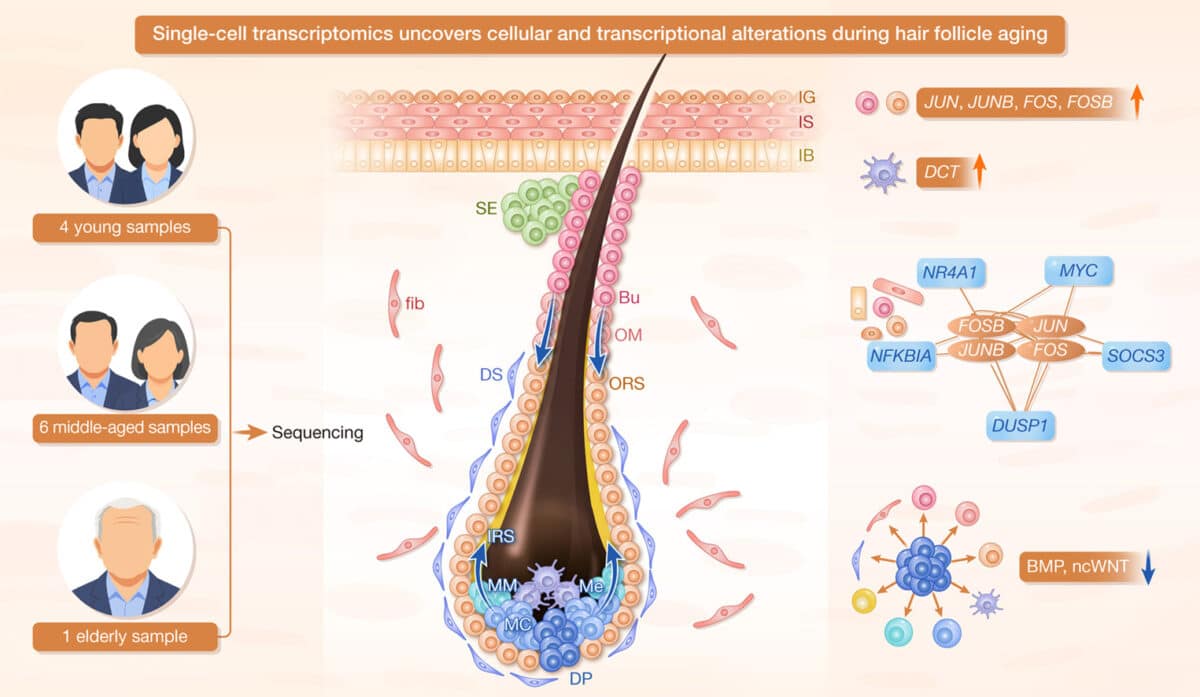
A team from BGI and Shanghai Skin Disease Hospital analyzed scalp samples from 11 individuals ranging from their 20s to late 50s, profiling the genetic activity of over 57,000 individual cells. For most analyses, the team compared a young-adult group with a midlife group. Their findings, published in hLife, reveal inflammation-linked gene activity across many cell types in middle-aged scalps, from the stem cells that generate new hair to the pigment-producing cells that determine hair color.
Hair follicle cells in people in their mid-30s to early 40s already display molecular signatures of stress, immune activation, and declining regenerative capacity—even when hair still appears relatively healthy.
Inflammation-Related Gene Patterns in Aging Hair
The research identified inflammation-related gene-expression patterns affecting many cell types in the hair follicle. Genes involved in stress responses, immune activation, and DNA damage were higher in middle-aged samples, while genes controlling stem cell renewal were suppressed. This creates conditions that may make it harder for hair follicle stem cells to function normally.
Central to this inflammatory pattern is a protein complex called AP-1. The researchers found markedly increased AP-1 activity in hair follicle stem cells and skin cells from middle-aged individuals. Network analysis suggests this protein complex coordinates other aging-related genes, including MYC and SOCS3. Based on prior work in other tissues, MYC can drive stem cells toward exhaustion through overactivity, while SOCS3 can block growth signals necessary for tissue regeneration. Together, these patterns point to a potential cycle of stem cell depletion.
Scientists call this broader process “inflammaging,” which is chronic, low-grade inflammation that increases with age throughout the body. This study demonstrates that the scalp shows similar gene-expression patterns.
Changes in Pigment-Producing Cells
Inflammation-related changes were particularly evident in melanocytes, the cells responsible for hair pigment. In middle-aged scalps, these cells showed elevated expression of DCT, a gene involved in melanin synthesis. Rather than indicating healthy pigment production, the authors interpret it as a sign pigment biology is being stressed or altered in midlife. How that connects to graying specifically still needs direct testing.
To validate their cellular findings, a follow-up group included 90 people who had trichoscopy, which measures hair density and diameter. Hair density declined with age across all groups, confirming that the molecular changes observed at the cellular level align with measurable physical changes.
When Cell Communication Weakens
Perhaps most revealing were the changes in how different cell types communicate with each other. The researchers analyzed molecular signals exchanged between cells and found overall communication decreased in middle-aged scalps.
Signaling pathways critical for hair growth cycles showed particularly notable changes. Dermal papilla cells, the specialized support cells at the base of each follicle, showed weakened communication with key follicle cells, including keratinocyte populations, in middle-aged samples. These signals normally tell follicles when to start growing new hair. When this communication breaks down, hair follicles may rest longer between growth cycles, potentially leading to progressively thinner hair.
The study also mapped three distinct populations of actively dividing cells in different scalp regions: the outer skin layer, the outer root sheath of hair follicles, and the hair matrix where new hair grows. These cells represent transitional stages between stem cells and fully differentiated hair cells, and their reduced numbers in middle-aged samples indicate impaired regenerative capacity.
Potential Targets for Future Research
The research could point to future therapeutic targets. If AP-1 activation and its downstream inflammatory patterns are part of what’s linked with hair follicle aging, interventions that modulate this pathway might one day help slow age-related hair changes, though this study didn’t test treatments. The molecular patterns in middle age may be modifiable. They involve specific cellular pathways that could, in theory, be targeted.
While the visible signs appear gradually, the underlying changes begin much earlier. This points to the possibility that interventions might be most effective when started in the 30s and 40s, before stem cell reserves are significantly depleted.
The researchers caution that their findings come from a limited sample size and don’t capture late-stage aging processes, since only one participant over 50 was included. The small sample size also prevented analysis of sex differences and regional variations across the scalp.
For the millions experiencing age-related hair changes, this research offers insight into the cellular patterns associated with hair aging and identifies molecular pathways that future treatments might target.
Disclaimer: This article provides general information about scientific research and is not medical advice. Consult a healthcare provider for concerns about hair loss or other health conditions.
Paper Summary
Limitations
The study acknowledges several constraints. The analysis included only one participant over 50, limiting conclusions about late-stage aging processes. The small sample size prevented comprehensive analysis of sex differences and regional variations across the scalp. Some transcriptional changes may follow non-linear patterns that only become apparent in advanced age, which this dataset could not capture. Additionally, while the research identified molecular associations with aging, it cannot definitively establish cause-and-effect relationships.
Funding and Disclosures
This research was supported by the Science, Technology, and Innovation Commission of Shenzhen Municipality (grant number JCYJ20220818102809021 to F.X.). The authors declare no competing financial interests or personal relationships that could influence the work reported.
Publication Details
Zhao Q, Ma R, Huang K, Wang J, Zhang D, Wang J, Ding X, Chen F, Zhao S, Ni N, Zhang X, Du Q, Lin X, Wan H, Zhang J, Ding X, Yang S, Xu F, Lai Y. “Single-cell RNA sequencing profiles age-related transcriptional landscapes in human hair follicle cells,” published in hLife, December 2025;3:626-646. DOI: 10.1016/j.hlife.2025.10.003. The corresponding authors are affiliated with BGI Cell (Shenzhen, Guangdong, China) and the Department of Dermatologic Surgery, Shanghai Skin Disease Hospital, Tongji University School of Medicine (Shanghai, China).







