
(© Natalie Board - stock.adobe.com)
LA JOLLA, Calif. — Studies show that women are more likely than men to develop Alzheimer’s, the most common form of dementia. Now, new findings focusing on the biological causes behind the disease may provide some much-needed insight into the reasons for this gender disparity. It turns out the higher risk could be linked to menopause.
A team from Scripps Research and Massachusetts Institute of Technology (MIT) discovered that an especially harmful, chemically modified form of an inflammatory immune protein called complement C3 was present in much higher levels in the brains of women who died with Alzheimer’s, in comparison to men who died with the disease.
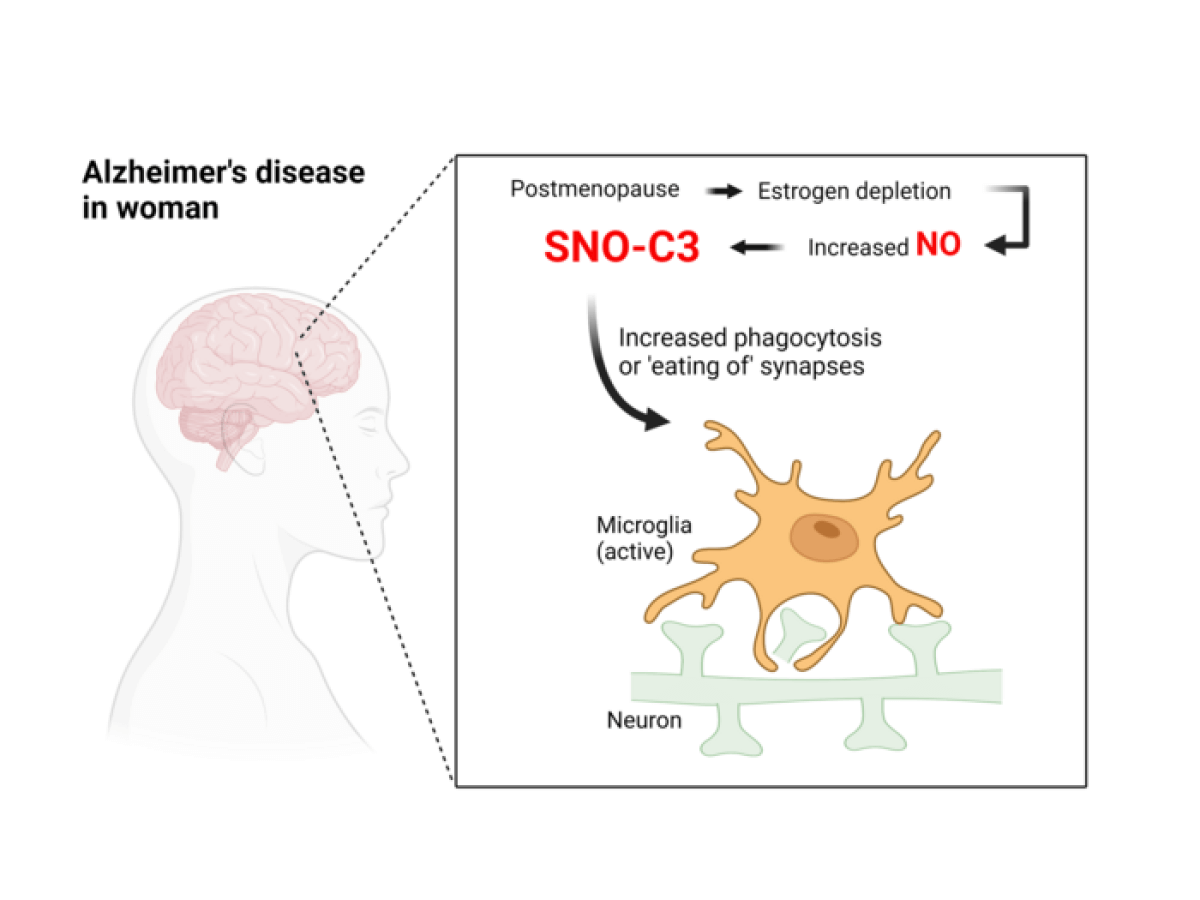
Importantly, study authors also say that estrogen normally protects against the development of this form of complement C3. Women’s estrogen levels naturally decline with age and as menopause sets in.
“Our new findings suggest that chemical modification of a component of the complement system helps drive Alzheimer’s, and may explain, at least in part, why the disease predominantly affects women,” says study senior author Stuart Lipton, MD, PhD, the Step Family Foundation Endowed Chair in the Department of Molecular Medicine at Scripps Research and a clinical neurologist in La Jolla, California, in a media release.
Alzheimer’s could be the results of a ‘SNO-storm’
Roughly six million Americans have a clinically diagnosed case of Alzheimer’s. Such a diagnosis is always fatal, with death usually occurring within a decade of onset. Even worse, there is currently no approved treatment that can slow the disease’s progression, let alone reverse the neurological damage it inflicts. Why can’t modern medicine develop something more effective? At the end of the day, scientists have never fully understood how Alzheimer’s develops, or why women account for nearly two-thirds of cases for that matter.
Dr. Lipton’s lab focuses on the study of biochemical and molecular events that may underlie neurodegenerative diseases, including the chemical reaction that forms a modified type of complement C3 — a process known as protein S-nitrosylation. Previously, Dr. Lipton’s team discovered this chemical reaction, which occurs when a nitric oxide (NO)-related molecule binds tightly to a sulfur atom (S) on a specific amino acid building block of proteins to form a modified “SNO-protein.”
Protein modifications by small clusters of atoms such as NO are actually quite common in cells, and usually serve to either activate or deactivate a target protein’s functions. S-nitrosylation, on the other hand, has been much harder for scientists to study due to technical issues. Still, Dr. Lipton suspects that “SNO-storms” of these proteins may be a major element of Alzheimer’s and other neurodegenerative disorders.
What’s triggering the destruction of brain connections?
For this latest research, study authors utilized novel methods for detecting S-nitrosylation to quantify proteins modified in 40 postmortem human brains. Half of those brains were from people who died of Alzheimer’s, while the other half were from people who died from other causes. Researchers divided the groups equally between men and women.
A total of 1,449 different proteins that had been S-nitrosylated were found in those brains. Among the most modified proteins, the team noted several that are tied to Alzheimer’s, including complement C3. Incredibly, S-nitrosylated C3 (SNO-C3) levels were more than six times higher among female brains with Alzheimer’s in comparison to male Alzheimer’s brains.
The complement system, an older evolutionarily part of the human immune system, consists of a family of proteins (including C3) that can activate one another to drive inflammation. Scientists call this process the “complement cascade.”
Studies over the last three decades have shown that in comparison to neurologically normal brains, Alzheimer’s brains have higher levels of complement proteins and other markers of inflammation. More recent research reveals that complement proteins can trigger brain-resident immune cells called microglia to destroy synapses, the vital connection points by which neurons send signals to one another. Currently, many scientists theorize this synapse-destroying mechanism at least partly drives the progression of Alzheimer’s disease. Moreover, loss of synapses has been shown to be a significant factor in cognitive decline among Alzheimer’s brains.
So, why would SNO-C3 be more common in female brains with Alzheimer’s?
Studies have long suggested that the female hormone estrogen can have brain-protective effects under certain conditions. Consequently, researchers hypothesize estrogen specifically protects women’s brains from C3 S-nitrosylation, but this protection weakens when estrogen levels fall sharply during menopause. Experiments conducted using cultured human brain cells supported this hypothesis and revealed that SNO-C3 increases as estrogen (β-estradiol) levels fall, thanks to the activation of an enzyme that makes NO in brain cells. The consequent increase in SNO-C3 activates microglial destruction of synapses.
“Why women are more likely to get Alzheimer’s has long been a mystery, but I think our results represent an important piece of the puzzle that mechanistically explains the increased vulnerability of women as they age,” Dr. Lipton concludes.
Moving forward, researchers plan to conduct further experiments with de-nitrosylating compounds — which remove the SNO modification. They hope to reduce pathology in animal models of Alzheimer’s and eventually among living humans.
The study is published in the journal Science Advances.







