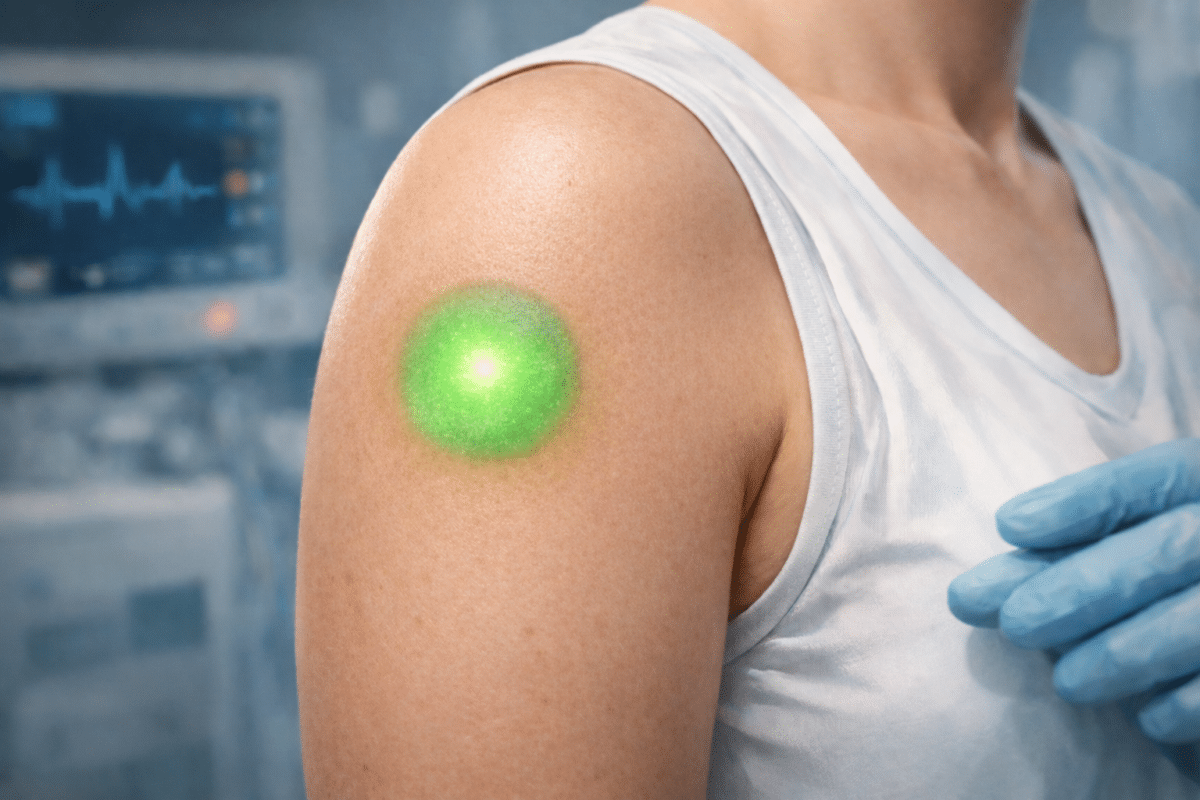
Concept illustration of a skin-implanted “Living Sensor Display,” designed so engineered keratinocyte stem cells produce a green fluorescent signal that can be observed from the skin surface when TNF-α–related inflammatory signaling is present. In the mouse experiments, fluorescence increased after TNF-α administration and peaked around day 1–2. (Image generated by OpenAI)
Green means go…to the doctor
In A Nutshell
- Scientists engineered living skin that glows green when it detects inflammation in the body, creating a biological early warning system for disease.
- The skin patch, transplanted onto mice, worked for over 200 days without batteries or maintenance; stem cells naturally renewed themselves and kept the sensor functioning.
- The tissue responded repeatedly to inflammation, lighting up within 24 hours each time researchers triggered immune responses, then fading as inflammation resolved.
- While human applications are years away, the technology could eventually shift medicine from treating diseases after symptoms appear to catching problems while they’re still reversible.
Scientists have created living skin that lights up bright green when inflammation rises inside the body. The engineered tissue, about the size of a quarter, works like a biological smoke detector, and is designed for long-term monitoring of inflammatory signals that often build silently before symptoms appear.
Researchers in Japan genetically modified human skin stem cells to glow when exposed to inflammatory molecules that surge during immune responses. They transplanted patches of this engineered skin onto mice, where it successfully integrated and kept working for over six months. Each time researchers triggered inflammation, the patch glowed brighter.
The breakthrough, published in Nature Communications, offers something current technology hasn’t been able to deliver: extended, real-time monitoring of inflammation within the body. While wearable devices track heart rate or steps, they can’t detect the inflammatory processes that drive many chronic diseases developing quietly over time.
Cells That Talk Back
The key was choosing the right type of cell. Skin stem cells naturally sit in the deepest layer of skin, churning out fresh skin cells throughout your lifetime. Scientists tweaked these cells so they’d produce a fluorescent protein whenever inflammatory signals hit their surface.
Getting the genetics right took some trial and error. After testing five different designs, researchers landed on a formula that produced the strongest glow. In lab dishes, modified cells started lighting up within 12 hours of exposure to inflammatory molecules. Higher inflammation meant brighter glow, showing the system could detect different levels of inflammatory activity.
The team built actual skin tissue by layering the modified cells together with other skin components. Within days, the lab-grown tissue developed multiple layers that looked and acted like real skin.
Living Sensors Beat Technology
Here’s why living cells work better than artificial devices: they’re extraordinarily sensitive. Inflammatory molecules like TNF-α exist in tiny amounts and break down within 15 minutes in the bloodstream. Blood tests can miss these fleeting signals entirely unless you happen to draw blood at exactly the right moment.
Cells solve this problem by acting as biological amplifiers. When an inflammatory molecule binds to a receptor, it triggers a cascade of reactions inside the cell, with each step multiplying the signal until it’s strong enough to see. Think of it like a whisper that gets passed along and amplified until it becomes a shout.
The modified cells also showed selectivity. When researchers tested the cells in lab dishes with different substances, they responded to specific inflammatory signals while ignoring unrelated molecules, reducing false alarms.
Six Months of Continuous Function
When scientists transplanted the engineered skin onto mice, something noteworthy happened. The tissue survived and matured. The initially thin patch thickened to match normal skin and even developed structural features resembling human skin that aren’t found in mice.
The patch stayed put for over 200 days, maintaining a stable size and location. The stem cells had settled in and were doing what stem cells do: continuously dividing and creating fresh cells that inherited the genetic modifications. No batteries, no external maintenance required.
Two months after transplantation, researchers tested whether the patches still worked. They injected inflammatory molecules near the transplant sites and watched. Within 24 hours, the patches glowed brighter. Peak brightness came on day one or two, then gradually faded as inflammation resolved, which is exactly what you’d want in an early warning system.
Four weeks later, they tested again. The patches responded just as strongly, proving they could detect inflammation repeatedly over time.
Inflammation Tracking Could Help Prevent Diseases
The team pushed further, testing whether the patches could catch inflammation triggered by the immune system itself. When they injected bacterial components into the mice, the patches didn’t respond directly to the bacteria. Instead, they detected inflammatory molecules released by the mice’s immune cells as they reacted to the threat.
This means the system can monitor the body’s overall immune response, not just one specific molecule. Interestingly, the second time researchers triggered immune activation, the glow was stronger. The paper notes this might be due to quirks in the particular mice used (which had partially compromised immune systems), but it raises intriguing questions about whether such patches could potentially track how the immune system responds to repeated challenges.
The researchers envision this approach as a way to monitor inflammation continuously over extended periods. Inflammation plays a role in many chronic diseases that develop over time, often progressing silently before causing symptoms.
If this technology could be adapted for humans, it might help catch inflammatory processes earlier in their development. The concept could extend beyond inflammation. Researchers suggest that modifying cells to respond to different signals could allow detection of hormones, stress markers, or metabolic changes.
The study authors describe their vision this way: “This Living sensor display holds promise for advancing health monitoring and disease prevention strategies by enabling continuous, sensitive, and specific in vivo biomarker tracking.”
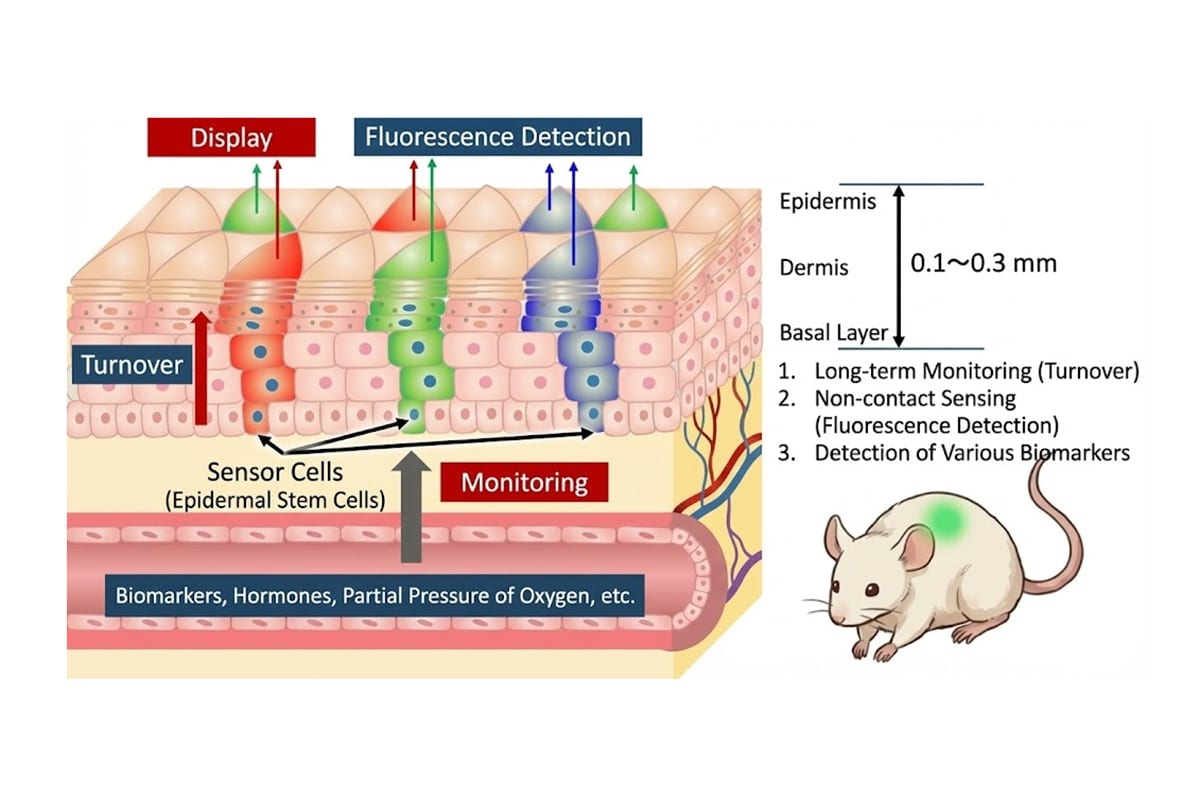
When Will The Living Skin Patch Be Available?
The current study used mice with compromised immune systems, which don’t fully represent how normal immune responses work. Testing in animals with intact immunity will be necessary to see if transplanted tissue faces rejection or causes unwanted inflammation itself. The patches required anesthesia for fluorescence measurements, limiting real-time monitoring in active animals.
Smaller patches produced weaker signals, setting a practical minimum size for detection. The fluorescent protein itself can sometimes trigger immune responses, potentially limiting how long patches function in recipients with normal immunity.
The mouse experiments demonstrated continuous function for over 200 days. Whether such patches could work for extended periods in humans remains to be tested, but the self-renewing nature of stem cells suggests long-term function may be possible.
Human applications remain distant, but veterinary medicine might adopt the technology sooner. Monitoring inflammation in valuable livestock or detecting chronic conditions in pets that can’t communicate their symptoms could provide early testing grounds.
For people, the concept of implanting genetically modified living tissue raises questions about long-term safety and societal acceptance. But the potential payoff (detecting inflammation as it develops rather than after it causes harm) makes the research worth pursuing. A biological monitor that alerts when inflammation rises, powered by your own metabolism and maintaining itself through natural cell turnover, could potentially shift how medicine approaches disease prevention.
This article is for informational purposes only and is not intended as medical advice.
Paper Notes
Limitations
The research used SCID Hairless Outbred (SHO) mice lacking mature B and T lymphocytes, which don’t fully represent normal immune responses in animals with complete immune systems. Although the team observed some adaptive immune characteristics after repeated LPS administration (possibly due to “leaky” SCID mice that develop limited lymphocyte function), testing in immunocompetent animals remains necessary. The engineered tissue responded to inflammatory signals but measurements required anesthetizing animals, limiting real-time monitoring in freely moving subjects. Fluorescence detection sensitivity decreased with smaller transplant sizes, as reduced tissue area produced weaker total signals. The study focused on a single biomarker pathway (NF-κB signaling), and expanding to additional targets requires further validation. Enhanced green fluorescent protein (EGFP) can trigger immune responses in some contexts, potentially limiting long-term viability in immune-competent hosts.
Funding and Disclosures
This work received support from JSPS KAKENHI Grant Numbers JP22H00284 and JP21H05013. Several authors (H. Fujita, J. Sawayama, M. Takeo, Y. Takayama, M. Ogawa, K. Yano, T. Tsuji, and S. Takeuchi) have filed a patent related to this work: Japanese Patent Application JP2023-141483, filed August 31, 2023. The remaining authors declared no competing interests. Y. Takayama and K. Yano are affiliated with Canon Medical Systems Co., a commercial entity that may have interests in biomedical technology development.
Publication Details
Authors: Jun Sawayama, Makoto Takeo, Yuki Takayama, Miki Takase, Hoshimi Aoyagi, Aki Takimoto, Saki Mizutani, Maiko Onuki, Satoshi Ikeo, Shogo Nagata, Miho Ogawa, Koji Yano, Takashi Tsuji, Shoji Takeuchi, and Hiroyuki Fujita
Affiliations: Institute of Industrial Science, The University of Tokyo; Graduate School of Information Science and Technology, The University of Tokyo; RIKEN Center for Biosystems Dynamics Research; Canon Medical Systems Co.; Tokyo City University
Journal: Nature Communications | Publication Date: January 12, 2026 | Volume/Issue: Volume 17, Article 56 | DOI: 10.1038/s41467-025-67384-2 | Article Type: Original Research Article | Manuscript History: Received May 16, 2024; Accepted November 28, 2025; Published online January 12, 2026 | Corresponding Author: Hiroyuki Fujita, email: hfujita@tcu.ac.jp







