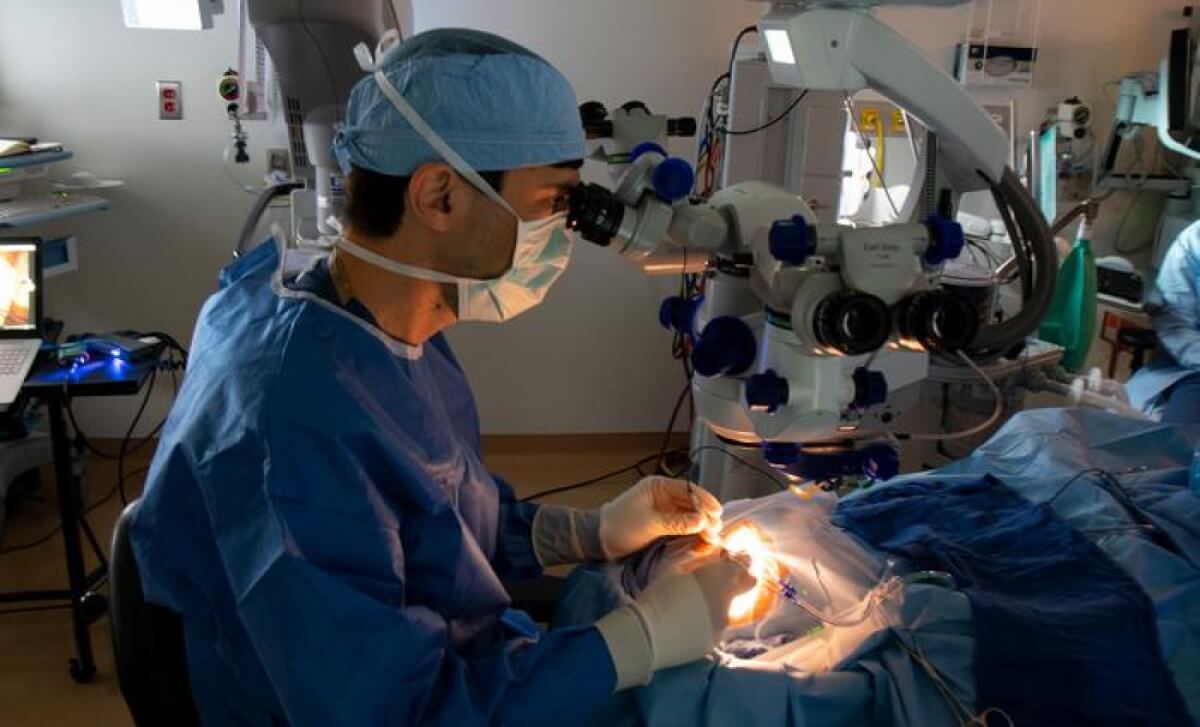
Jason Comander, MD, PhD, performs the procedure to deliver the CRISPR-based medicine as part of the BRILLIANCE trial in September 2020 at Mass Eye and Ear. (CREDIT: Mass Eye and Ear)
BOSTON — For children born with a rare form of childhood blindness called Leber congenital amaurosis (LCA) often caused by a single genetic mutation, an innovative new treatment using CRISPR gene editing technology is showing promising results in restoring vision.
LCA is a genetic disorder that affects the retina, the light-sensitive tissue at the back of the eye responsible for detecting light and color. Children with LCA often experience severe vision loss or complete blindness from a very young age. One of the most common causes of LCA is a defect in the CEP290 gene, which plays a crucial role in the development and function of the retina’s light-sensing cells, called photoreceptors.
Until now, there have been no effective treatments for LCA resulting from CEP290 mutations. However, the new study led by researchers at Massachusetts Eye and Ear and Oregon Health & Science University offers a glimmer of hope. Published in the New England Journal of Medicine, researchers tested a novel therapy called EDIT-101 in 14 patients with LCA.
So, what exactly is EDIT-101, and how does it work? In simple terms, it’s a gene editing tool that uses CRISPR technology to correct the genetic defect in CEP290. CRISPR, which stands for “clustered regularly interspaced short palindromic repeats,” is a powerful gene editing technique that allows scientists to make precise changes to DNA.
You can think of CRISPR as a pair of molecular scissors that can cut out a defective piece of DNA and replace it with a corrected version. In the case of EDIT-101, the CRISPR tool is designed to specifically target and repair the most common mutation in the CEP290 gene that causes LCA.
Here’s how the treatment works: The EDIT-101 complex, which contains the CRISPR components, is injected directly into the retina of the patient’s worse-seeing eye in a surgical procedure. Once inside the retina, the CRISPR tool goes to work, finding and cutting out the CEP290 mutation and allowing the gene to function normally. The goal is to restore the activity of the photoreceptor cells and improve vision.
In the study, 14 LCA patients age three and older received a single injection of EDIT-101 at three different dose levels. The patients were then followed for up to two years to assess the safety and effectiveness of the treatment.
The results, while preliminary, are encouraging. No serious side-effects related to the treatment were observed, suggesting that EDIT-101 is safe. More excitingly, 11 out of the 14 patients (79%) showed some improvement in at least one measure of visual function, such as visual acuity, retinal sensitivity to light, navigational abilities, or quality of life scores.
Six patients (43%) had meaningful improvements in cone photoreceptor function, the cells responsible for color vision and visual acuity. This is especially noteworthy because LCA primarily affects rod photoreceptors that are responsible for night and peripheral vision, while cone cells are relatively spared. Enhancing cone function could potentially have a big impact on patients’ central vision and daily functioning.
“There is nothing more rewarding to a physician than hearing a patient describe how their vision has improved after a treatment,” says Dr. Mark Pennesi of the Casey Eye Institute at Oregon Health & Science University, where the first in vivo CRISPR treatment took place, in a media release. “One of our trial participants has shared several examples, including being able to find their phone after misplacing it and knowing that their coffee machine is working by seeing its small lights. While these types of tasks might seem trivial to those who are normally sighted, such improvements can have a huge impact on quality of life for those with low vision.”
The youngest participants, two children who were nine and 14 years-old, seemed to have the most robust response, with both showing improvements in multiple visual outcomes. This suggests that intervening early, while the retina is still developing, may yield the greatest benefits.
While these initial results are promising, it’s important to remember that this is still an early-stage trial with a small number of patients. Larger and longer studies will be needed to fully assess the safety and efficacy of EDIT-101. Scientists will also need to fine-tune the treatment to optimize the dose and timing of the injections for maximum benefit.
Nonetheless, the fact that a single injection of EDIT-101 could lead to measurable improvements in vision in these patients with previously untreatable blindness is a major milestone. It provides proof-of-concept that CRISPR gene editing can be successfully used to treat inherited retinal diseases, paving the way for its application to other forms of blindness and potentially other genetic disorders.
For families affected by LCA, this research represents a beacon of hope. The possibility of restoring even a small amount of vision could have a profound impact on the lives of these children, allowing them to better navigate their environment, perform daily activities, and experience the world in a whole new light.
Of course, there are still many questions to be answered and challenges to overcome before CRISPR therapies like EDIT-101 become widely available. However, this study is a crucial first step in demonstrating the feasibility and promise of this cutting-edge approach.
In the grand scheme of things, the implications of this research extend far beyond just LCA. CRISPR gene editing has the potential to revolutionize the treatment of countless genetic diseases that have long been considered incurable. By correcting the underlying genetic defects, we may one day be able to not just manage the symptoms of these disorders but actually cure them at their source.
For the children and families affected by LCA, that day can’t come soon enough. With each passing breakthrough, the light at the end of the tunnel grows a little brighter, offering renewed hope for a future where blindness is no longer a life sentence but a treatable condition. And that is truly a vision worth fighting for.







