As of April 16, 2024, there have been 19 reports of individuals experiencing adverse reactions to what they thought were Botox injections, according to the U.S. Food and Drug Administration. Instead, these patients received substances described as misbranded, from unlicensed sources, adulterated, counterfeit, contaminated, improperly stored and transported, unsafe, and ineffective.
Nine people who received counterfeit Botox were hospitalized, and four needed treatment with botulism antitoxin due to concerns that the botulinum toxin had spread beyond the injection site.
The symptoms these patients experienced included:
- Double or blurred vision
- Drooping eyelids
- Difficulty swallowing
- Dry mouth
- Slurred speech
- Fatigue
- Generalized weakness
They are the same symptoms as botulism and call for emergency attention. If botulism goes untreated, it can progress to paralysis, respiratory failure, and death. According to the Cleveland Clinic, botulism antitoxin will arrest the effects of the toxin but will not reverse the damage already done. There may be lingering weakness, shortness of breath, and nervous system symptoms.
The states that have reported counterfeit Botox incidents include Colorado, Florida, Illinois, Kentucky, Nebraska, New Jersey, New York, Tennessee, and Washington.
According to Michelle Waltenberg, senior botulism epidemiologist at the Centers for Disease Control and Prevention (CDC), the sources of these products are unknown, which is a matter of great concern. Waltenberg is working with the FDA and state health departments in the investigation to identify the source(s) of the counterfeit Botox.
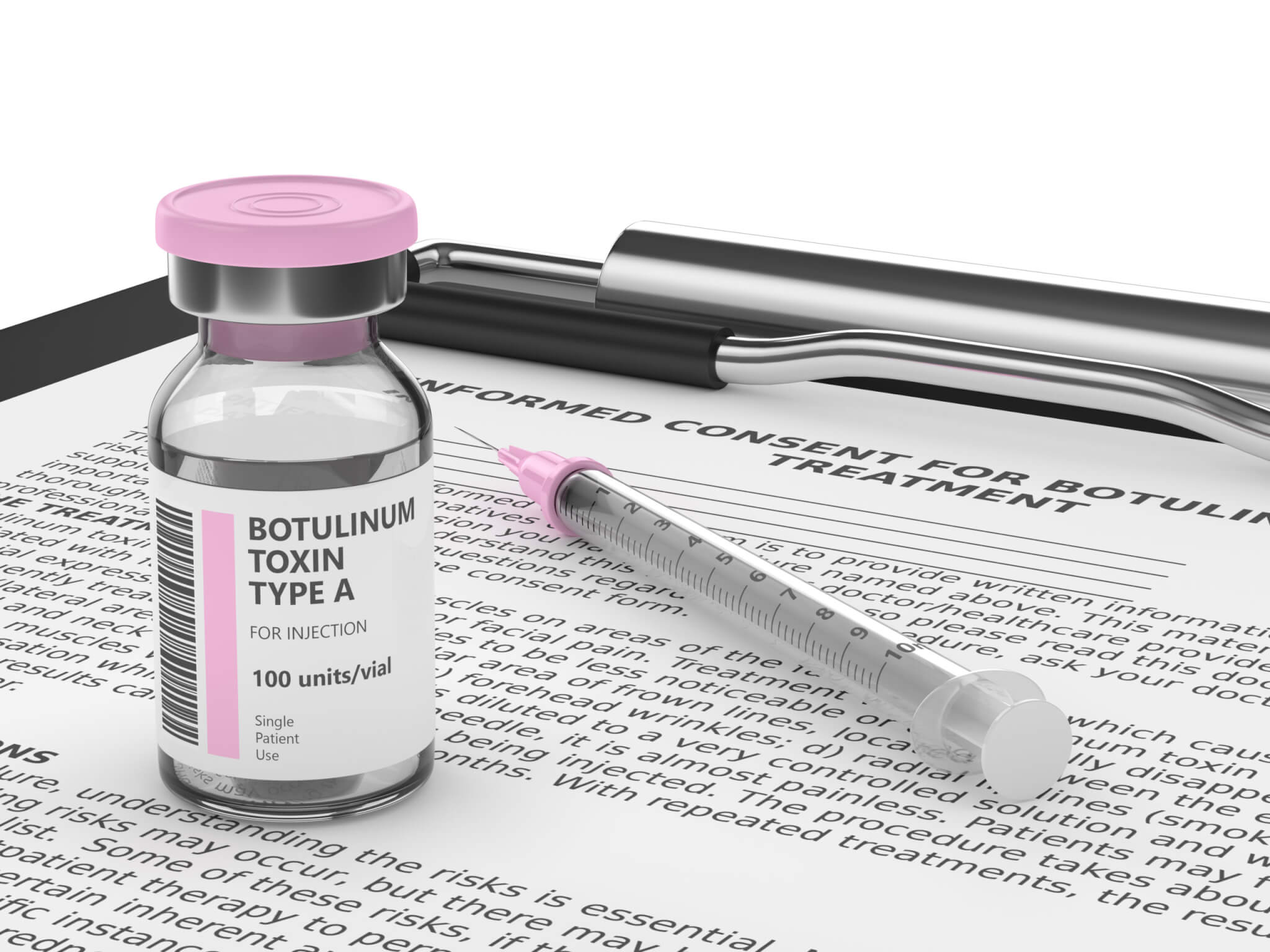
How to tell real from fake Botox
AbbVie manufactures authentic Botox products in 50, 100, and 200-unit dose forms. The product descriptions on the outer cartons are either “BOTOX® COSMETIC / onabotulinumtoxinA / for Injection” or “OnabotulinumtoxinA / BOTOX® / for injection.”
The manufacturer identified on the fakes’ outer carton is either “Allergan Aesthetics / An AbbVie Company” or “abbvie.” FDA-approved Botox displays the active ingredient as “OnabotulinumtoxinA” on the outer carton and vial.
Currently, there is no indication that the reported events were linked to AbbVie’s FDA-approved Botox, and the genuine product should be considered safe and effective for its intended and approved uses.
The counterfeit product includes counterfeiting of the outer carton and vial. The counterfeit product may be identified by one or more of the following:
- The outer carton and vial contain lot number C3709C3
- The outer carton displays the active ingredient as “Botulinum Toxin Type A” instead of “OnabotulinumtoxinA”
- The outer carton and vial indicate 150-unit doses, which is not a unit dose made by AbbVie or Allergan
- The outer carton contains language that is not English
If you’re considering getting Botox, the CDC recommends asking your provider if they are licensed and trained to give Botox injections. Some states have a license look-up tool where you can check. You should also ask if their botulinum toxin is approved by the FDA and obtained from a reliable source.
Consumers should report suspected counterfeit Botox products to the FDA at 800-551-3989, or through www.accessdata.fda.gov/scripts/email/oc/oci/contact.cfm.
Healthcare professionals and consumers should report adverse events related to the use of any medications, including suspected counterfeit medications, to FDA’s MedWatch Safety Information and Adverse Event Reporting Program:
- Complete and submit the report online at MedWatch Online Voluntary Reporting Form
- Download and complete the form, then submit it via fax at 1-800-FDA-0178