
Brown fat tissue burns calories rather than storing them (Photo by Towfiqu Barbhuiya on Unsplash)
UPPSALA, Sweden — Even our immune systems tend to pack on a few extra pounds as we grow older. Now, researchers from Uppsala University in Sweden are detailing why human lymph nodes tend to gradually accumulate fat and lose their function with age, as well as how this influences the effectiveness of our immune system.
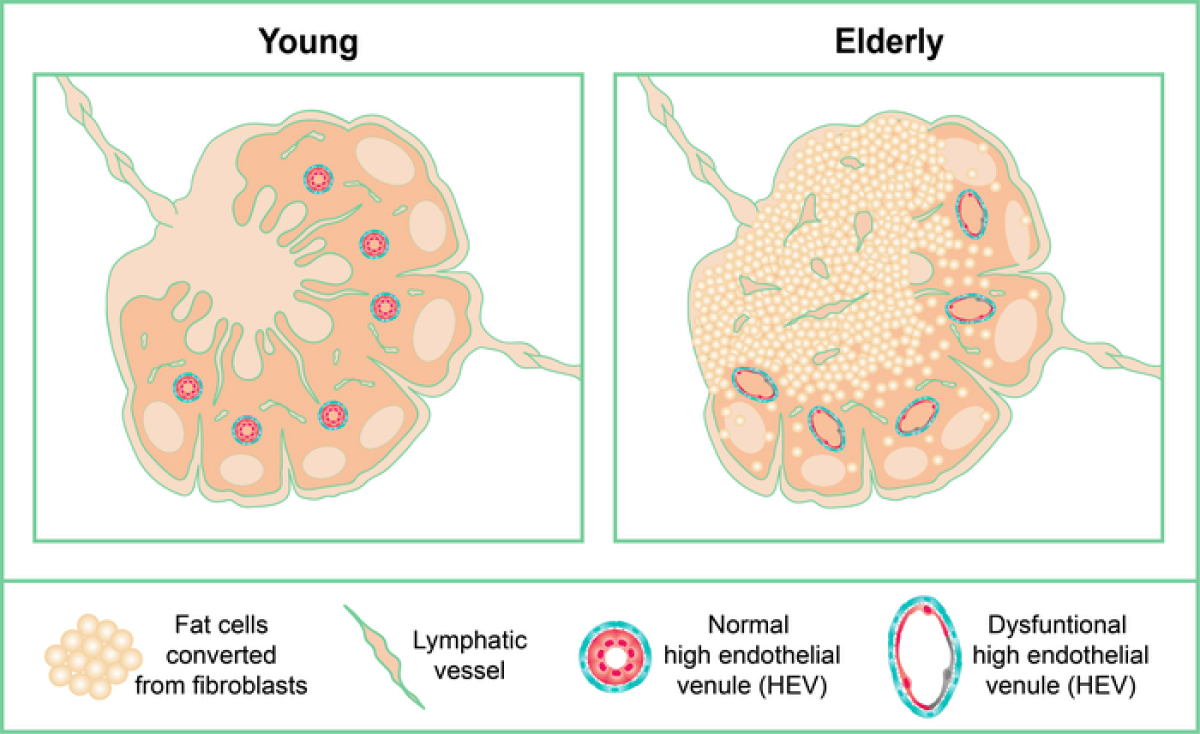
Lymph nodes are small bean-shaped structures which scientists consider the “headquarters” of the human immune system. Whenever someone develops an infection, for example, the lymph nodes are the sites where immune cells congregate, activate, and proliferate to put together an effective immune defense. However, as a person grows older, studies show the normal tissues in lymph nodes (the stroma) tend to gradually disappear, replaced by adipose tissue (fat). This is called lymph node lipomatosis, and, while it is very common and increases with age, there has been little research into understanding lipomatosis.
Study authors analyzed more than 200 lymph nodes, demonstrating that lipomatosis begins in the central part of the lymph node — the medulla. They also collected evidence linking lipomatosis to the transformation of the supporting cells of lymph nodes (fibroblasts) into adipocytes (fat cells). Notably, specific types of fibroblasts located in the medulla were especially prone to becoming adipocytes.
Even during the early stages of lipomatosis, researchers noted negative changes that hindered the lymph nodes’ abilities to provide effective immunity. More specifically, the specialized blood and lymphatic vessels that normally provide channels for immune cells to enter and exit the lymph node were destroyed in parts of the node where fat had formed.
Consequently, researchers theorize lipomatosis of lymph nodes, even at early stages, may be an important factor in the documented poorer response to vaccinations observed in elderly people. Eventually, fat consumes the entire lymph node, rendering it totally unable to function.
“Our study is a first step towards understanding why lipomatosis occur, and towards the longer term goal of finding ways to prevent its progression and the destruction of the lymph node,” says Tove Bekkhus, first author of the study, in a media release.
Animal studies fail to replicate human lymph nodes
Animal models are frequently used to study the effects of aging, but researchers say as of now they cannot mimic these observed effects in human lymph nodes in animal models. This highlights the importance of research conducted using humans when it comes to studying the effects of aging.
“I hope our work will spur an interest among other researchers in including lymph nodes lipomatosis as a factor when studying elderly people’s responses to vaccination and infections. The changes we observe are also highly relevant to cancer research, since in several types of cancer, the lymph nodes are the first place cancer cells spread to,” explains study leader Maria H. Ulvmar, a researcher at Uppsala University.
“Our publication provides the first chapter of a story about fat and loss of function in our lymph nodes when we age. We will now continue to develop this story by designing new studies to learn more about the underlying causes and consequences of these changes,” Ulvmar continues.
The main samples analyzed for this project were provided by the Uppsala Biobank. Those samples were analyzed via advanced image analysis. This project also included analysis and experiments using cell cultures from primary stromal cells and bioinformatic analysis of gene expressions (RNA level) taken from two single cell RNA sequencing (RNAseq) data sets, mouse and human, previously published but analyzed for the first time in this study.
The study is published in The Journal of Pathology.







