SAN FRANCISCO — The fight against Alzheimer’s disease has taken a promising turn as the U.S. Food and Drug Administration (FDA) is gearing up to approve the third in a new series of drugs to fight the condition.
While encouraging strides have been made, experts emphasize that the best outcomes for patients come when doctors detect the disease in its earliest stages.
This is likely “just the opening chapter in a new era of molecular therapies for Alzheimer’s disease and related neurodegenerative disorders,” says Dr. Gil Rabinovici, director of the University of California-San Francisco Alzheimer’s Disease Research Center, in a commentary published in JAMA.
Donanemab, a monoclonal antibody similar to its predecessors, aducanumab (Aduhelm) and lecanemab (Leqembi), targets plaques in the brain formed by the protein amyloid. The accumulation of these plaques impedes cellular function and promotes the spread of another protein, tau. Both proteins play significant roles in the onset of Alzheimer’s disease.
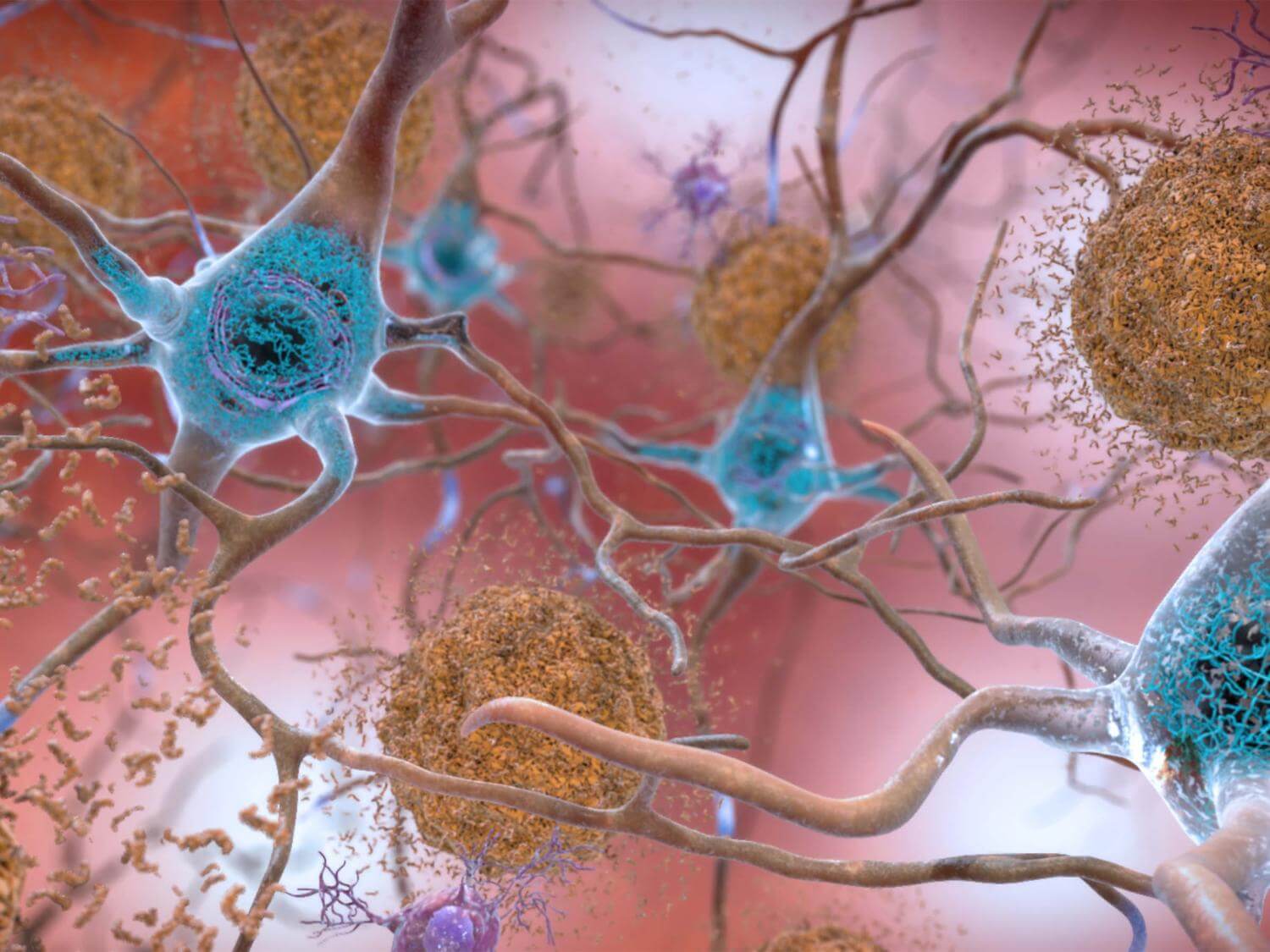
The recent trial reveals that donanemab can reduce cognitive decline by 35 percent compared to a placebo in patients showing low-to-intermediate tau levels in their brains. This drug also presents a 40-percent decreased risk of patients progressing from mild cognitive impairment to mild dementia or from mild-to-moderate dementia. Notably, donanemab outperforms its counterparts in clearing amyloid plaques, though it doesn’t significantly affect tau concentrations in crucial brain areas.
Despite these promising findings, Dr. Rabinovici emphasizes the need for a deeper analysis to truly comprehend the implications on patient outcomes.
The study also highlighted the limitations of donanemab. Patients in advanced disease stages didn’t benefit significantly compared to those on the placebo. Coupled with potential severe side-effects, Dr. Rabinovici stressed the need to “aim higher in developing more impactful and safer treatments.”
Of concern, donanemab is associated with ARIA (amyloid-related imaging abnormalities), which may lead to brain swelling and microbleeds. Serious ARIA symptoms manifested in 3.7 percent of patients, even resulting in three deaths. Especially at risk are those with the APOE4 gene, linked to a heightened Alzheimer’s risk. With that in mind, Dr. Rabinovici recommends genetic testing before treatment and advises that medical practitioners exercise caution when administering these drugs.
A notable flaw in the trial was its lack of racial and ethnic diversity, with only 8.6 percent of the 1,251 U.S. participants being non-White. Dr. Rabinovici also raised ethical questions, pointing out the higher dementia rates among Black and Latino populations and questioning the trial’s broad applicability.
Due to the high cost of donanemab and the expected surge in demand, Dr. Rabinovici suggested limiting treatment duration solely to the time required to clear amyloid plaques.
According to Dr. Rabinovici, this could “greatly enhance the feasibility of treatment for patients, clinicians, insurers, and health systems.”
You might also be interested in:
- What is Leqembi? Here’s why the FDA just approved this drug as an Alzheimer’s treatment
- A minty cure? Smelling menthol reduces symptoms of Alzheimer’s disease
- Scientists develop most accurate way of identifying dementia risk

First, lots of 90-somethings in the Leisure World Study have brains loaded with beta-amyloid plaques, but no global neuronal cell loss of dementia. A Dutch study last year confirmed those results. So is beta-amyloid causative or a tag along.
Second, none of these antibodies have demonstrated superiority to memantine-ER. Show us a head to head comparison of proposed drugs with daily oral 28 mg memantine-ER. If they cannot prove superiority to a drug with much less risk, why should anyone prescribe it?