On July 17, the U.S. Food and Drug Administration (FDA) approved Beyfortus (nirsevimab-alip), the first and only drug for the prevention of respiratory syncytial virus (RSV) lower respiratory tract disease in newborns and infants during or entering their first year of this seasonal condition. It is also for children up to 24 months of age who are at risk for severe RSV disease or through their second RSV season.
“RSV can cause serious disease in infants and some children and results in a large number of emergency department and physician office visits each year,” says John Farley, M.D., M.P.H., director of the Office of Infectious Diseases in the FDA’s Center for Drug Evaluation and Research, in a statement. “Today’s approval addresses the great need for products to help reduce the impact of RSV disease on children, families, and the health care system.”
RSV is an extremely common cause of acute respiratory infection in individuals of all ages. Virtually all children have the illness by their second birthday. Most of these young children have mild, cold-like symptoms. Some infants, however, usually with their first infection, develop lower respiratory tract diseases such as pneumonia or bronchiolitis.
Some conditions cause a child to be at greater risk for severe RSV infection. These include:
- Mother smoking during pregnancy
- Premature birth, especially before 29 weeks gestation
- Chronic lung disease of prematurity
- Low birth weight infants
- Some heart defects present at birth
- Weak immune system due to other illnesses or their treatment
- Having siblings
- Exposure to secondhand smoke in their home
- History of eczema
- History of allergies
- Exposed to children in a childcare setting
- Crowded living conditions
Bronchiolitis is the more common complication of severe RSV illness. The small breathing tubes of the lungs (bronchioles) swell, blocking airflow through the lungs. Infants develop bronchiolitis more often than older children because their airways are smaller and more easily blocked.
Bronchiolitis often starts with the signs you expect with a cold, such as a runny nose, mild cough, and fever. In one or two days, the cough may get worse, and an infant will begin to breathe faster. If your child shows signs of trouble breathing, call your child’s doctor or go to an emergency room. The signs of trouble breathing include:
- Widening nostrils and squeezing the muscles under the rib cage, trying to get more air in and out of the lungs.
- Grunting and tightening their stomach muscles.
- Making a high-pitched whistling sound, called wheezing, when they are trying to breathe out.
- Trouble drinking because they may have trouble sucking and swallowing.
- If it gets very difficult for your baby to breathe, you may notice a bluish tint around their lips and fingertips, meaning they are not getting enough oxygen.
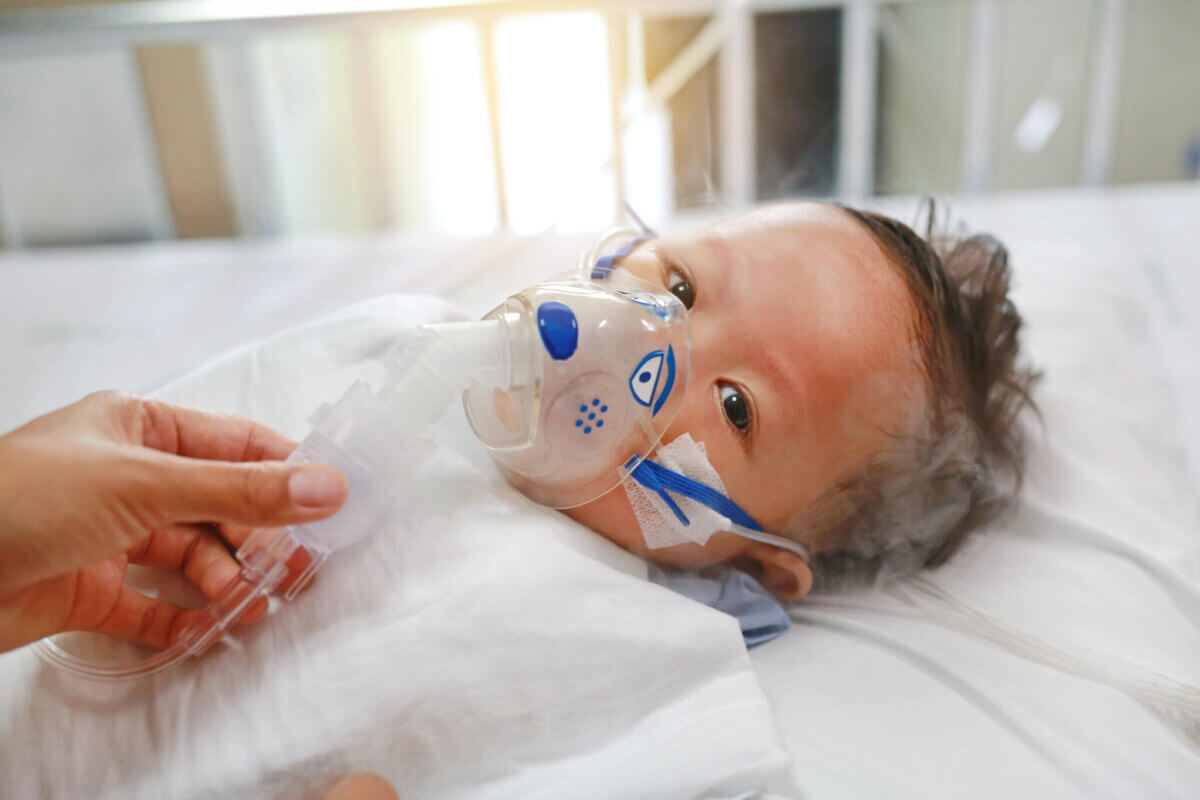
Approximately one to three percent of children under 12 months of age in the United States are hospitalized each year due to RSV, according to the American Academy of Pediatrics. They may require oxygen, IV fluids (if they are not eating and drinking), and/or a machine (ventilator) to help with breathing. Most improve quickly with this supportive care.
In most regions of the U.S., RSV illness has been seasonal, with cases predominantly in the late fall, winter, and early spring. With mask-wearing, isolation, and physical distancing for COVID-19 management, there were fewer cases of RSV in 2020. Once safety measures relaxed with the arrival of COVID-19 vaccines, however, a rise in RSV cases began in spring 2021, surging in the summer. RSV is not adhering as closely to its previous annual pattern of case occurrences, leading to the use of the term “interseasonal” rather than seasonal.
Beyfortus is a monoclonal antibody. Monoclonal antibodies are laboratory-made proteins that fight off harmful causes of infectious diseases, such as viruses. They mimic the body’s natural immune system. One dose of Beyfortus, as a single intramuscular injection before or during RSV season, may protect during the RSV season.
The safety and effectiveness of Beyfortus were supported by three clinical trials (Trials 03, 04, and 05).
Trial 03 included 1,453 preterm infants born during or entering their first RSV season. Of the 1,453 preterm infants in the trial, 969 received a single dose of Beyfortus and 484 received a placebo. Among infants treated with Beyfortus, 2.6 percent experienced RSV lower respiratory tract infection, compared with 9.5 percent of infants who received a placebo (an inactive solution.) Beyfortus reduced the risk of RSV lower respiratory tract infection by about 70 percent relative to placebo.
In Trial 04, Beyfortus reduced the risk of RSV lower respiratory tract infection by approximately 75 percent relative to placebo.
Trial 05 was a randomized, double-blind, multicenter trial. It supported using Beyfortus in children who remain at risk for severe RSV disease up to their second birthday or second RSV season.
Possible side-effects of Beyfortus include injection site reactions and rash. Beyfortus comes with warnings about severe hypersensitivity reactions, including life-threatening anaphylaxis. Caution must be used when administering Beyfortus to infants and children with clinically significant bleeding disorders.
The FDA granted Beyfortus, manufactured by AstraZeneca, a Fast Track designation for this indication. A children’s vaccine for RSV is in development.