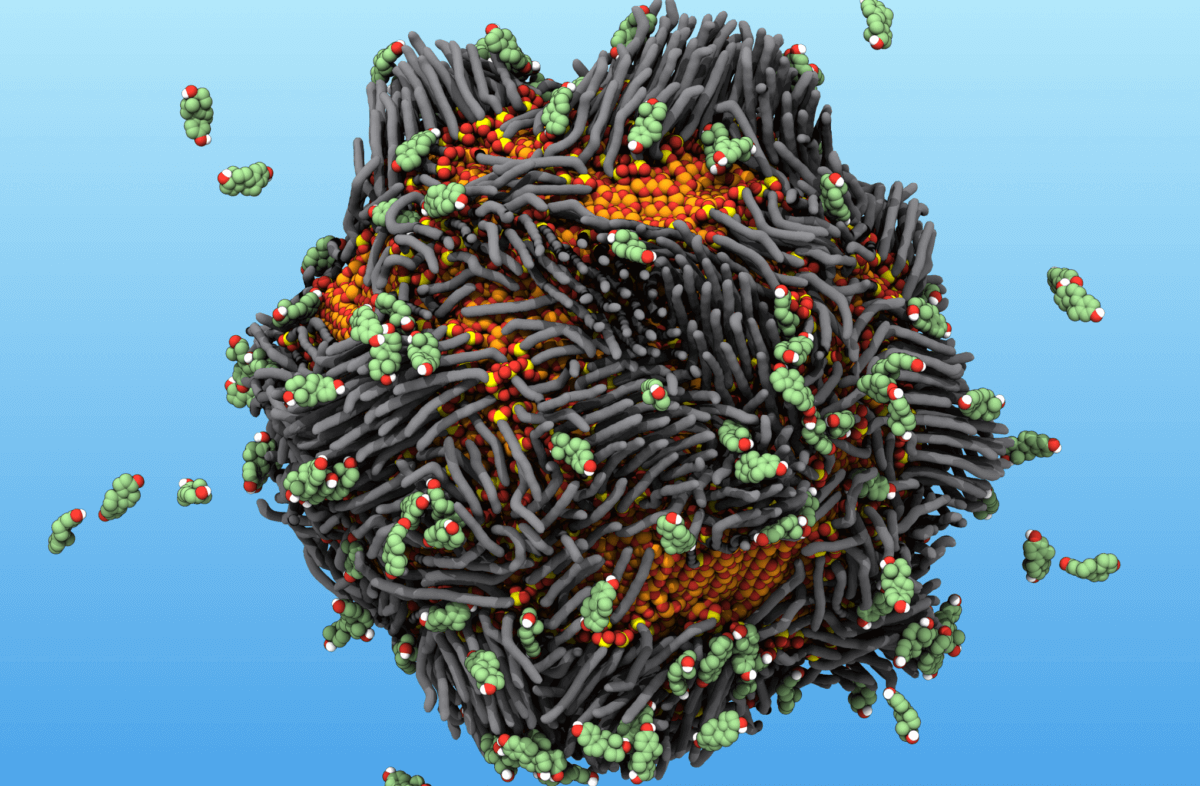
In this illustration, a “smart rust” nanoparticle attracts and traps estrogen molecules, which are represented by the floating objects. (Credit: Dr. Dustin Vivod and Prof. Dr. Dirk Zahn, Computer Chemistry Center (CCC), Friedrich-Alexander-Universität Erlangen-Nürnberg)
SAN FRANCISCO — Iron rust is generally considered a nuisance, but German researchers have now transformed it into a water-cleaning superhero they aptly name “smart rust.” This innovative particle is not only magnetic but can also pull in numerous pollutants like oil, tiny plastic particles, and even the herbicide glyphosate.
Perhaps its most remarkable feat? It can capture potentially harmful estrogen hormones, addressing environmental concerns related to aquatic life.
“Our ‘smart rust’ is cheap, nontoxic and recyclable,” says Dr. Marcus Halik, the project’s principal investigator from the Friedrich-Alexander-Universität Erlangen-Nürnberg, in a statement. “And we have demonstrated its use for all kinds of contaminants, showing the potential for this technique to improve water treatment dramatically.”
How does the smart rust work?
At the heart of “smart rust” are iron oxide nanoparticles, which are attracted to magnets but won’t stick to each other, ensuring they remain dispersed. To amplify their pollution-grabbing potential, the researchers coat these particles with phosphonic acid molecules, turning the particles into tiny pollutant sponges.
“After we add a layer of the molecules to the iron oxide cores, they look like hairs sticking out of these particles’ surfaces,” says Dr. Halik.

By tweaking the other end of these acid molecules, they can customize the particles to pull in various pollutants.
The team’s earlier versions were successful in clearing crude oil from Mediterranean Sea water and glyphosate from pond water. Additionally, these nanoparticles proved capable of removing minute plastic particles from lab and river water samples.
Scroll down to see video on how smart rust nanoparticles clean water
But the team wasn’t stopping there. Lukas Müller, a graduate student associated with the project, explored whether this rust could tackle trace pollutants, specifically hormones. Estrogens, which are hormones naturally found in humans and livestock, often find their way into water sources and can disrupt the ecosystem, even in minuscule amounts. “I started with the most common estrogen, estradiol, and then four other derivatives that share similar molecular structures,” says Müller.
Müller’s approach involved crafting “pockets” on the smart rust’s surface that attract and lock in the estradiol. Though these pockets are too small to be seen, advanced instruments are helping Müller confirm their existence and effectiveness.
“We are trying to use different puzzle pieces to understand how the molecules actually assemble on the nanoparticles’ surface,” explains Müller.
The next steps involve testing these particles in real-world scenarios and assessing their reusability. With their high surface area and numerous “pockets”, these particles promise effective cleaning over multiple uses. “By repeatedly recycling these particles, the material impact from this water treatment method could become very small,” notes Dr. Halik.
The research was presented during the fall meeting of the American Chemical Society.







