NEW YORK — Your nose is made up of countless cells, and a new study has found that each of those cells detects different smells. Scientists from Columbia’s Zuckerman Institute say the mammalian nose, a marvel of evolution, is equipped with millions of nerve cells, each with a unique receptor for detecting specific odor chemicals. This intricate system allows us to identify a trillion distinct scents, influencing everything from what people choose to eat, to recognizing friends or foes and triggering old memories.
“How sensory cells in the nose make their receptor choices has been one of the most vexing mysteries about olfaction,” says Stavros Lomvardas, PhD, a Roy and Diana Vagelos Professor and Chair of Biochemistry and Molecular Biophysics and Herbert and Florence Irving Professor of Neuroscience at Columbia’s Zuckerman Institute and the Vagelos College of Physicians and Surgeons, and corresponding author of the study. “Now, the story behind our sense of smell, or olfaction, is becoming clearer, and also more dramatic.”
For instance, some neurons in our noses are exclusively sensitive to ethyl vanillin, the primary odorant in vanilla, while others are tuned to detect limonene, the main scent in lemons.
Dr. Lomvardas described the process as a dramatic “Squid Games-style” competition occurring within the tiny space of each neuron’s nucleus, where chromosomes and genes reside. Here, a multitude of olfactory receptor genes compete against each other, eventually narrowing down to a single winner that determines the cell’s sensitivity to a specific odorant.
“It’s basically a battle between 1,000 contenders,” explains Ariel Pourmorady, the paper’s first author and an MD-PhD candidate at the Zuckerman Institute in the Lomvardas lab, in a university release.

This complex process involves various gene-regulating molecules that either promote or inhibit the production of olfactory receptors. These molecules form alliances to turn specific genes on or off.
Dr. Lomvardas’s team also discovered a portion of the genome he calls the “Greek Islands,” named for their resemblance to islands in the Aegean Sea. These molecular hubs reshape parts of the genome, influencing which receptor genes are favored.
While RNA is commonly known for translating genetic code into proteins, such as those detecting odorants, this study suggests it also plays a key part in altering the genome’s structure. This alteration enhances the expression of one olfactory receptor gene while suppressing others.
“It turns out that the genome has a certain spatial organization in the nucleus and changes in this structure are pivotal when it comes to which genes are expressed into proteins, like olfactory receptors,” says Pourmorady. “We are learning just how important this process is within maturing olfactory cells.”
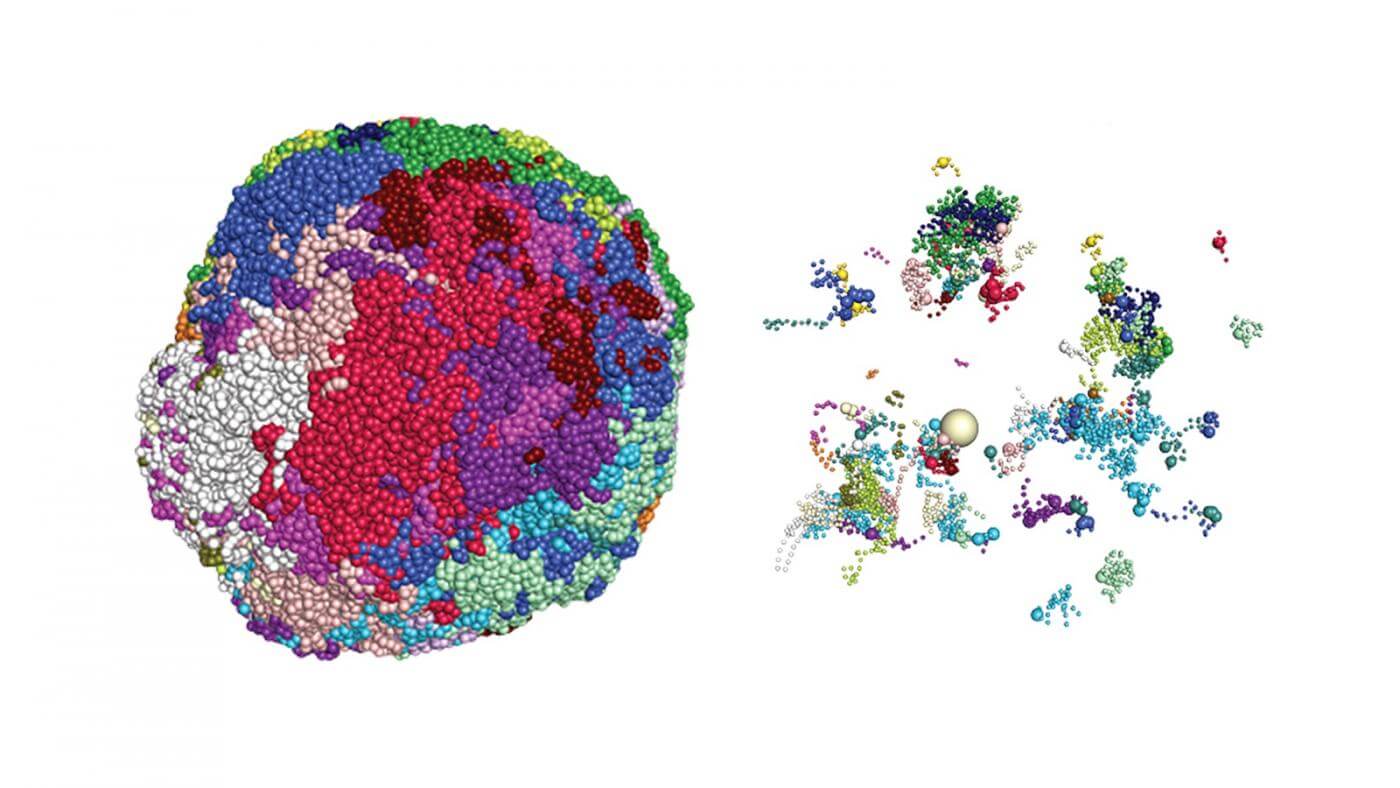
Although there are still unanswered questions, the researchers have outlined a process that starts with maturing olfactory cells expressing multiple receptor genes. Eventually, RNA narrows these down to a single gene. The RNA produced at the most active hub wins the competition, boosting receptor-gene expression. Interestingly, RNA from the same hub may travel to other hubs, causing structural changes in the genome that suppress gene expression. The end result is a nose filled with mature olfactory neurons, each dedicated to detecting only one specific scent.
“We are reaching the edge of science fiction when it comes to the molecular and genomic details we now can observe inside a single cell’s nucleus,” Dr. Lomvardas concludes. “We need to keep going back in to figure out the rest of this olfaction puzzle.”
The findings are published in the journal Nature.
You might also be interested in:
- Follow your nose! Study finds people choose friends based on how they smell
- Why do some farts smell and some don’t? And why do some farts feel hot?
- Smell of science: Researchers develop first-ever robot able to sniff odors

