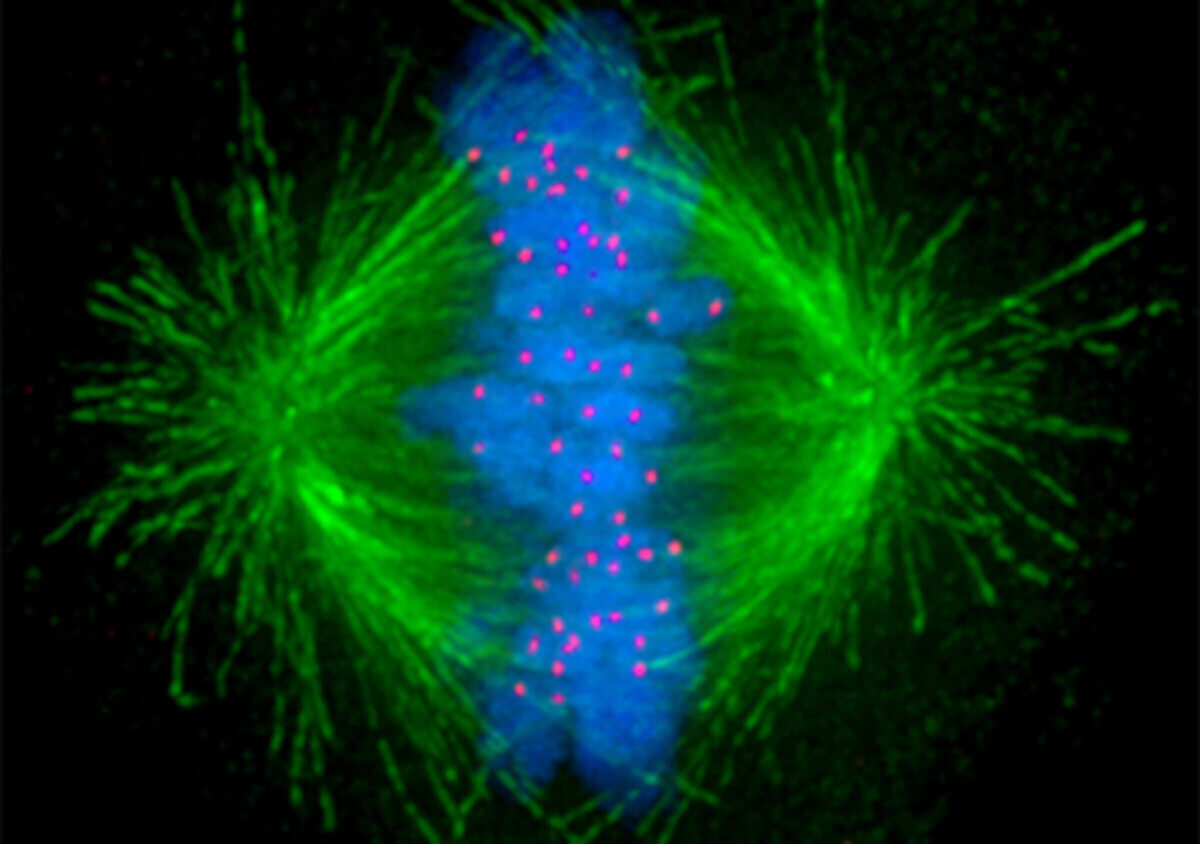
Tiny tube-like structures inside the cell called microtubules (in green) that help separate chromosomes evenly during cell division can overcome low exposures to chemotherapy. (Image: Wikimedia Commons/University of New South Wales)
BIRMINGHAM, United Kingdom — Cancer is a complex disease with a knack for eluding treatment strategies, but a recent study has revealed how to combat this. Australian researchers have discovered how cancer cells thwart a common cancer therapy and, more importantly, how this knowledge could pave the way for targeted drugs to battle it.
The study conducted by the University of New South Wales (UNSW) exposes how cancer cells employ a force-generating rescue mechanism to stabilize a critical cell structure responsible for cell division, thus resisting the effects of chemotherapy.
“We discovered that cancer cells use the mechanical force supplied by the edge of the cell called the cell cortex, to overcome the impact of commonly used chemotherapy that blocks the ability of the cell to separate the chromosomes during cell division,” says study senior author Peter Gunning, professor at the School of Biomedical Sciences, UNSW Medicine & Health, in a university release. “Now that we understand this exact pathway cancer cells use to avoid the toxic effects of the chemotherapy, it opens the door to improving cancer treatments.”
The chemo-defeating mechanism revealed
During cell division, or mitosis, microtubules, which are tiny tube-like structures inside the cell, act as “arms” responsible for separating genetic material evenly, ensuring the successful production of daughter cells. Since cancer cells undergo rapid division, targeting these structures using anti-microtubule chemotherapy drugs is a common strategy to inhibit their growth.
“The anti-microtubule chemotherapy usually fragments the mechanical arms into multiple hubs that pull the chromosomes in multiple directions rather than the normal two. The resulting chaos prevents proper separation of the chromosomes to the two daughter cells and induces apoptosis, or programmed cell death,” notes Gunning. “At high doses of chemotherapy this is quite effective. But at lower exposure, which is a core problem, particularly as you get to the interior of solid tumors or when chemotherapy doses must be reduced due to toxicity in patients, cancer cells activate a rescue mechanism.”
However, cancer cells have a clever escape plan. When exposed to lower doses of chemotherapy, often required in the core of solid tumors or when reduced due to patient toxicity, cancer cells activate a rescue mechanism. This mechanism arises because cancer cells trigger a signal recognizing microtubule fragmentation, causing the arms to reach out toward the cell’s edge, pulling on the cortex to reunite the fragments.
“It was quite surprising to us because we did not expect this mechanism of the cancer cell to be used in this way to overcome the cancer therapy, but we could see it happening before our eyes.” Prof. Gunning says.
“This allows the arms to stabilize and generate the force necessary to physically grab and pull the chromosomes into each daughter cell and ensure the cancer cell multiplies.”

This surprising discovery emerged from noticing a specific drug combination that improved chemotherapy for neuroblastoma, a childhood cancer. To comprehend how it worked, advanced imaging was employed to observe the mechanism in real time during cell division.
Researchers believe this mechanism might be a fundamental aspect of cell biology, serving as a fallback mechanism that allows any cell to overcome minor microtubule disruptions to ensure its survival.
The next step for the researchers is to develop drugs that work in conjunction with current chemotherapy agents to overcome the cancer cell resistance mechanism. Before advancing to clinical studies, they plan to enhance the drugs’ activity in animal models over the next few years.
“By attacking the force-generating machinery built by the cancer cells we expect that we will be able to allow the cancer therapy to do its job much more effectively,” concludes Gunning. “In practical terms, we have established a company that will allow us to develop the drugs needed to attack this rescue mechanism, enabling the anti-microtubule chemotherapy to act more effectively, and hopefully improve patient outcomes.”
The study is published in the journal Current Biology.







