
(© Casimiro - stock.adobe.com)
CAMBRIDGE, United Kingdom — A new treatment using stem cell injections may halt the progression of multiple sclerosis (MS), an innovative study suggests. Collaborative research between European and American scientists shows this treatment could safeguard the brains of MS patients from further harm. In pioneering human clinical trials, patients who received stem cell injections showed no escalation in disability or symptom severity.
The study raises hopes for additional trials that could lead to treatments for progressive MS, which affects over two million people worldwide. Although current treatments can reduce relapse severity and frequency, about two-thirds of MS patients still experience a transition to a debilitating progressive phase within 25 to 30 years of diagnosis.
What is multiple sclerosis?
MS occurs when the immune system attacks the myelin sheath protecting nerve fibers, disrupting communication within the brain and spinal cord. Macrophages, key immune cells, typically remove harmful agents from the body but can harm the central nervous system (CNS) in progressive MS, causing chronic inflammation and nerve damage.
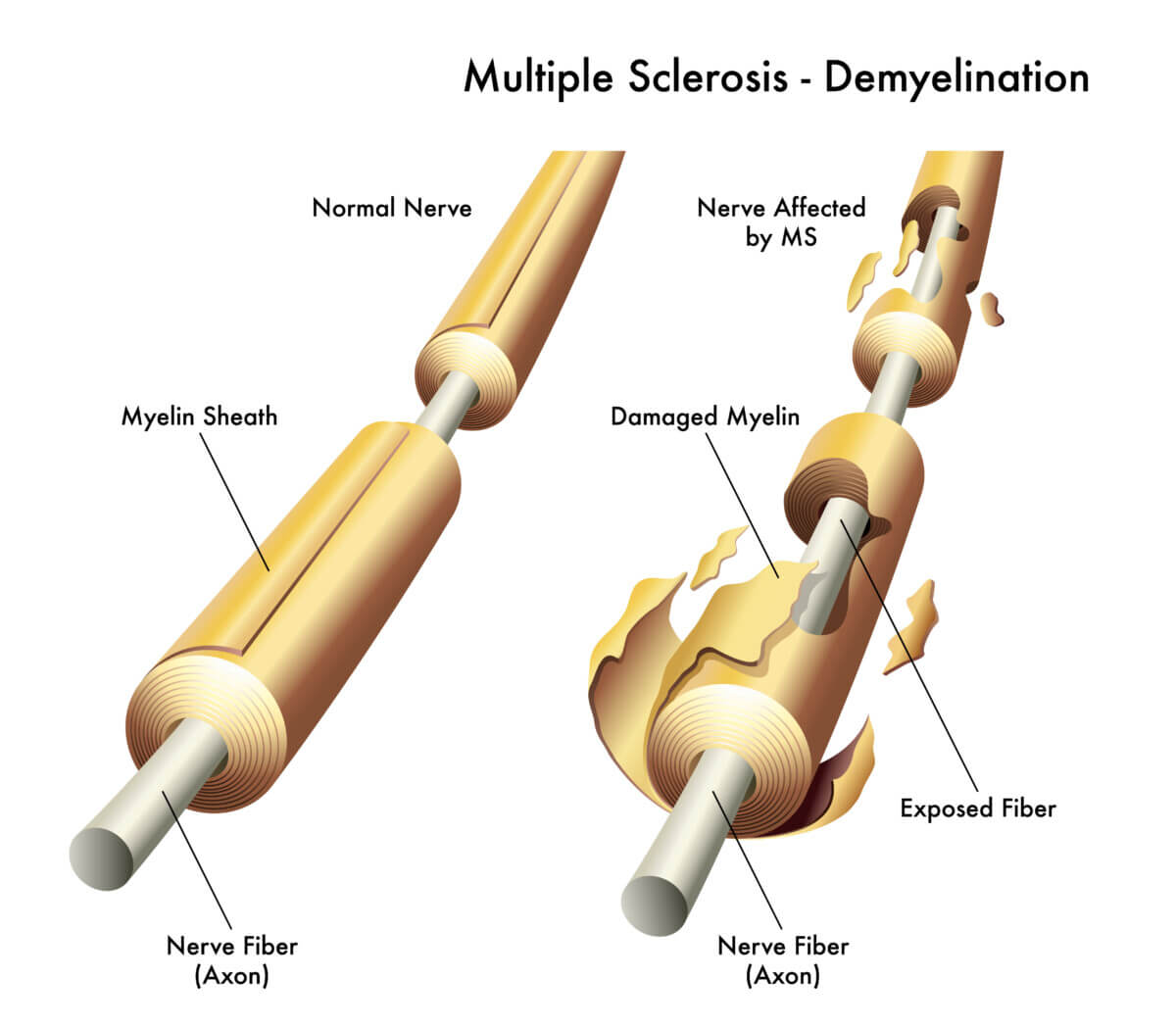
Recent advancements in stem cell transplantation have increased the anticipation for new therapies to mitigate this damage. These “master cells” can transform into almost any cell type in the body.
University of Cambridge researchers previously showed that skin cells, reprogrammed as brain stem cells and transplanted into the CNS, can reduce inflammation and potentially repair MS damage in mice.
The latest study, including teams from the United Kingdom, United States, Switzerland, and Italy, conducted a landmark early-stage clinical trial. They injected neural stem cells into the brains of 15 secondary MS patients from two Italian hospitals. The stem cells, derived from a single miscarried fetal donor, could potentially be replaced by cells from patients themselves in the future, resolving issues related to using fetal tissue.
Over the following year, the 15 patients were monitored, with no severe adverse events reported. Despite high levels of disability at the trial’s onset, with most participants wheelchair-bound, no patient showed increased disability or symptom deterioration.
Interestingly, a higher stem cell dose correlated with a lesser reduction in brain tissue volume, suggesting an anti-inflammatory effect. The study also observed signs of a neuroprotective impact, with altered brain metabolism potentially reprogramming microglial cells.
Higher doses also led to increased and sustained levels of fatty acids, associated with the treatment’s effectiveness and disease progression.
Despite the study’s limitations, including its small scale and potential effects of immunosuppressant drugs, the findings are exceptionally promising, according to Professor Stefano Pluchino of the University of Cambridge.
“We desperately need to develop new treatments for secondary progressive MS, and I am cautiously very excited about our findings, which are a step towards developing a cell therapy for treating MS,” says Prof. Pluchino in a university release.
The trial’s success supports proceeding to more extensive clinical trials.
“It has taken nearly three decades to translate the discovery of brain stem cells into this experimental therapeutic treatment,” adds Professor Angelo Vescovi of the University of Milano-Bicocca. “This study will add to the increasing excitement in this field and pave the way to broader efficacy studies, soon to come.”
The team says their trial demonstrates that the stem cell injections are safe and might stabilize disability progression. They add further clinical trials are essential to determine the treatment’s efficacy.
The study is published in the journal Cell Stem Cell.
You might also be interested in:
- Over-the-counter allergy medicine shows promise in potentially reversing multiple sclerosis
- More fiber may help fight multiple sclerosis, study reveals
- Revolutionary 3D-Printing Technique Uses Stem Cells To Repair Brain Injuries
South West News Service writer James Gamble contributed to this report.







